
Rice Science ›› 2023, Vol. 30 ›› Issue (1): 70-86.DOI: 10.1016/j.rsci.2022.05.005
• Research Paper • Previous Articles
Chen Yanhua,#, Wang Yaliang,#, Chen Huizhe, Xiang Jing, Zhang Yikai, Wang Zhigang, Zhu Defeng, Zhang Yuping( )
)
Received:2022-01-17
Accepted:2022-05-18
Online:2023-01-28
Published:2022-11-11
Contact:
Zhang Yuping
About author:First author contact:#These authors contributed equally to this work
This is an open access article under the CC BY-NC-ND license (
Peer review under responsibility of China National Rice Research Institute
Chen Yanhua, Wang Yaliang, Chen Huizhe, Xiang Jing, Zhang Yikai, Wang Zhigang, Zhu Defeng, Zhang Yuping. Brassinosteroids Mediate Endogenous Phytohormone Metabolism to Alleviate High Temperature Injury at Panicle Initiation Stage in Rice[J]. Rice Science, 2023, 30(1): 70-86.
Add to citation manager EndNote|Ris|BibTeX
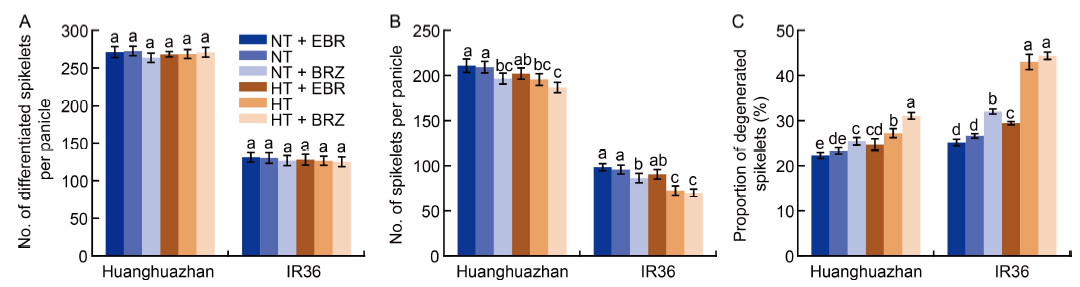
Fig. 1. Effects of exogenous 24-epibrassinolide (EBR) or brassinazole (BRZ) treatments on the number of differentiated spikelets per panicle (A), the number of spikelets per panicle (B) and proportion of degenerated spikelets (C) under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions. Values are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant differences between the treatments (P < 0.05).
| Cultivar | Treatment | No. of effective panicles per hill | Seed-setting rate (%) | Grain weight (mg) | Yield per hill (g) |
| Huanghuazhan | NT + EBR | 8.27 ± 0.32 a | 84.34 ± 0.82 a | 23.64 ± 0.41 a | 23.66 ± 0.24 a |
| NT | 8.60 ± 0.46 a | 83.75 ± 0.40 a | 23.34 ± 0.54 a | 22.43 ± 0.29 b | |
| NT + BRZ | 8.13 ± 0.21 a | 82.07 ± 0.64 b | 22.55 ± 0.27 bc | 20.74 ± 0.36 c | |
| HT + EBR | 8.20 ± 0.36 a | 83.52 ± 0.29 a | 23.02 ± 0.38 ab | 21.88 ± 0.33 b | |
| HT | 8.20 ± 0.36 a | 82.10 ± 0.48 b | 22.23 ± 0.23 c | 20.91 ± 0.57 c | |
| HT + BRZ | 8.23 ± 0.51 a | 81.06 ± 1.21 b | 21.12 ± 0.25 d | 18.88 ± 0.33 d | |
| IR36 | NT + EBR | 11.77 ± 0.38 a | 84.68 ± 0.75 a | 22.92 ± 0.35 a | 21.83 ± 0.24 a |
| NT | 11.77 ± 0.45 a | 83.48 ± 0.55 ab | 22.75 ± 0.39 a | 19.24 ± 0.95 b | |
| NT + BRZ | 11.74 ± 0.38 a | 80.84 ± 0.76 c | 22.49 ± 0.12 a | 17.91 ± 1.82 b | |
| HT + EBR | 12.17 ± 0.40 a | 82.73 ± 0.96 b | 21.35 ± 0.16 b | 18.19 ± 0.30 b | |
| HT | 11.87 ± 0.38 a | 81.18 ± 0.92 c | 19.99 ± 0.41 c | 13.89 ± 0.58 c | |
| HT + BRZ | 11.93 ± 0.32 a | 79.85 ± 0.51 c | 18.94 ± 0.46 d | 12.11 ± 0.31 d | |
| Values are Mean ± SD (n = 3). Different lowercase letters in the same column indicate significant differences between the treatments (P < 0.05). | |||||
Table 1. Effects of exogenous 24-epibrassinolide (EBR) or brassinazole (BRZ) treatments on rice yield and its components under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions.
| Cultivar | Treatment | No. of effective panicles per hill | Seed-setting rate (%) | Grain weight (mg) | Yield per hill (g) |
| Huanghuazhan | NT + EBR | 8.27 ± 0.32 a | 84.34 ± 0.82 a | 23.64 ± 0.41 a | 23.66 ± 0.24 a |
| NT | 8.60 ± 0.46 a | 83.75 ± 0.40 a | 23.34 ± 0.54 a | 22.43 ± 0.29 b | |
| NT + BRZ | 8.13 ± 0.21 a | 82.07 ± 0.64 b | 22.55 ± 0.27 bc | 20.74 ± 0.36 c | |
| HT + EBR | 8.20 ± 0.36 a | 83.52 ± 0.29 a | 23.02 ± 0.38 ab | 21.88 ± 0.33 b | |
| HT | 8.20 ± 0.36 a | 82.10 ± 0.48 b | 22.23 ± 0.23 c | 20.91 ± 0.57 c | |
| HT + BRZ | 8.23 ± 0.51 a | 81.06 ± 1.21 b | 21.12 ± 0.25 d | 18.88 ± 0.33 d | |
| IR36 | NT + EBR | 11.77 ± 0.38 a | 84.68 ± 0.75 a | 22.92 ± 0.35 a | 21.83 ± 0.24 a |
| NT | 11.77 ± 0.45 a | 83.48 ± 0.55 ab | 22.75 ± 0.39 a | 19.24 ± 0.95 b | |
| NT + BRZ | 11.74 ± 0.38 a | 80.84 ± 0.76 c | 22.49 ± 0.12 a | 17.91 ± 1.82 b | |
| HT + EBR | 12.17 ± 0.40 a | 82.73 ± 0.96 b | 21.35 ± 0.16 b | 18.19 ± 0.30 b | |
| HT | 11.87 ± 0.38 a | 81.18 ± 0.92 c | 19.99 ± 0.41 c | 13.89 ± 0.58 c | |
| HT + BRZ | 11.93 ± 0.32 a | 79.85 ± 0.51 c | 18.94 ± 0.46 d | 12.11 ± 0.31 d | |
| Values are Mean ± SD (n = 3). Different lowercase letters in the same column indicate significant differences between the treatments (P < 0.05). | |||||
| Treatment | BR content | aCTK content | Z + ZR content | iPA content | aGA content | GA1 + GA3 content | GA4 + GA7 content | IAA content | ABA content |
| Huanghuazhan | |||||||||
| NT + EBR | 3.47 ± 0.11 a | 14.89 ± 0.04 a | 5.59 ± 0.30 a | 9.30 ± 0.25 a | 13.02 ± 0.20 a | 5.39 ± 0.19 a | 7.63 ± 0.31 a | 47.73 ± 1.73 a | 57.22 ± 1.36 d |
| NT | 3.36 ± 0.01 ab | 13.62 ± 0.13 b | 5.30 ± 0.20 ab | 8.32 ± 0.31 b | 11.73 ± 0.23 b | 5.03 ± 0.27 a | 6.70 ± 0.06 b | 45.57 ± 1.41 ab | 57.26 ± 0.72 d |
| NT + BRZ | 3.25 ± 0.07 bc | 12.44 ± 0.08 c | 4.54 ± 0.10 c | 7.90 ± 0.05 bc | 9.39 ± 0.22 c | 4.96 ± 0.18 a | 4.44 ± 0.10 d | 41.57 ± 0.80 c | 62.41 ± 0.08 c |
| HT + EBR | 3.16 ± 0.08 cd | 13.46 ± 0.20 b | 5.25 ± 0.18 b | 8.21 ± 0.38 b | 11.64 ± 0.27 b | 5.34 ± 0.34 a | 6.30 ± 0.13 c | 45.39 ± 1.32 b | 76.89 ± 0.79 a |
| HT | 3.08 ± 0.08 d | 11.92 ± 0.24 d | 4.43 ± 0.10 c | 7.48 ± 0.19 cd | 8.72 ± 0.22 d | 4.46 ± 0.15 b | 4.25 ± 0.11 d | 44.87 ± 0.84 b | 70.29 ± 2.03 b |
| HT + BRZ | 2.67 ± 0.06 e | 11.01 ± 0.22 e | 3.74 ± 0.07 d | 7.27 ± 0.20 d | 8.34 ± 0.27 d | 4.50 ± 0.16 b | 3.84 ± 0.13 e | 40.91 ± 1.07 c | 62.37 ± 1.36 c |
| IR36 | |||||||||
| NT + EBR | 3.52 ± 0.14 a | 14.98 ± 0.29 a | 4.34 ± 0.05 a | 10.64 ± 0.26 a | 14.53 ± 0.01 a | 4.69 ± 0.14 a | 9.84 ± 0.15 a | 44.67 ± 1.48 a | 63.66 ± 1.62 b |
| NT | 3.21 ± 0.08 b | 12.44 ± 0.02 b | 4.07 ± 0.17 b | 8.38 ± 0.15 b | 9.27 ± 0.11 c | 4.65 ± 0.14 a | 4.62 ± 0.04 c | 44.88 ± 1.76 a | 57.27 ± 1.36 c |
| NT + BRZ | 2.88 ± 0.09 c | 10.85 ± 0.10 c | 3.77 ± 0.14 c | 7.08 ± 0.24 c | 8.05 ± 0.12 e | 4.08 ± 0.12 b | 3.97 ± 0.12 d | 43.75 ± 1.54 a | 55.72 ± 0.39 cd |
| HT + EBR | 3.20 ± 0.07 b | 12.32 ± 0.10 b | 4.10 ± 0.20 b | 8.23 ± 0.22 b | 9.74 ± 0.08 b | 4.62 ± 0.07 a | 5.12 ± 0.13 b | 41.50 ± 0.53 b | 70.73 ± 2.73 a |
| HT | 2.08 ± 0.08 d | 9.66 ± 0.26 d | 2.91 ± 0.09 d | 6.75 ± 0.17 c | 8.56 ± 0.12 d | 4.12 ± 0.09 b | 4.44 ± 0.21 c | 41.09 ± 0.47 b | 62.50 ± 0.57 b |
| HT + BRZ | 1.63 ± 0.01 e | 8.81 ± 0.09 e | 2.43 ± 0.05 e | 6.37 ± 0.13 d | 7.09 ± 0.21 f | 3.46 ± 0.13 c | 3.62 ± 0.08 e | 40.74 ± 0.33 b | 53.86 ± 1.05 d |
| BR, Brassinolide; Z, Zeatin; ZR, Zeatin riboside; iPA, N6-(Δ2-isopentenyl) adenosine riboside; aCTK, Active cytokinins (Z + ZR + iPA); GA1, Gibberellin A1; GA3, Gibberellin A3; GA4, Gibberellin A4; GA7, Gibberellin A7; aGA, Active gibberellins (GA1 + GA3 + GA4 + GA7); IAA, Indole-3-acetic acid; ABA, Abscisic acid. Values are Mean ± SD (n = 3). Different lowercase letters in the same column indicate significant differences between the treatments (P < 0.05). | |||||||||
Table 2. Effects of exogenous 24-epibrassinolide (EBR) or brassinazole (BRZ) treatments on phytohormone concentrations in rice panicles under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions. ng/g
| Treatment | BR content | aCTK content | Z + ZR content | iPA content | aGA content | GA1 + GA3 content | GA4 + GA7 content | IAA content | ABA content |
| Huanghuazhan | |||||||||
| NT + EBR | 3.47 ± 0.11 a | 14.89 ± 0.04 a | 5.59 ± 0.30 a | 9.30 ± 0.25 a | 13.02 ± 0.20 a | 5.39 ± 0.19 a | 7.63 ± 0.31 a | 47.73 ± 1.73 a | 57.22 ± 1.36 d |
| NT | 3.36 ± 0.01 ab | 13.62 ± 0.13 b | 5.30 ± 0.20 ab | 8.32 ± 0.31 b | 11.73 ± 0.23 b | 5.03 ± 0.27 a | 6.70 ± 0.06 b | 45.57 ± 1.41 ab | 57.26 ± 0.72 d |
| NT + BRZ | 3.25 ± 0.07 bc | 12.44 ± 0.08 c | 4.54 ± 0.10 c | 7.90 ± 0.05 bc | 9.39 ± 0.22 c | 4.96 ± 0.18 a | 4.44 ± 0.10 d | 41.57 ± 0.80 c | 62.41 ± 0.08 c |
| HT + EBR | 3.16 ± 0.08 cd | 13.46 ± 0.20 b | 5.25 ± 0.18 b | 8.21 ± 0.38 b | 11.64 ± 0.27 b | 5.34 ± 0.34 a | 6.30 ± 0.13 c | 45.39 ± 1.32 b | 76.89 ± 0.79 a |
| HT | 3.08 ± 0.08 d | 11.92 ± 0.24 d | 4.43 ± 0.10 c | 7.48 ± 0.19 cd | 8.72 ± 0.22 d | 4.46 ± 0.15 b | 4.25 ± 0.11 d | 44.87 ± 0.84 b | 70.29 ± 2.03 b |
| HT + BRZ | 2.67 ± 0.06 e | 11.01 ± 0.22 e | 3.74 ± 0.07 d | 7.27 ± 0.20 d | 8.34 ± 0.27 d | 4.50 ± 0.16 b | 3.84 ± 0.13 e | 40.91 ± 1.07 c | 62.37 ± 1.36 c |
| IR36 | |||||||||
| NT + EBR | 3.52 ± 0.14 a | 14.98 ± 0.29 a | 4.34 ± 0.05 a | 10.64 ± 0.26 a | 14.53 ± 0.01 a | 4.69 ± 0.14 a | 9.84 ± 0.15 a | 44.67 ± 1.48 a | 63.66 ± 1.62 b |
| NT | 3.21 ± 0.08 b | 12.44 ± 0.02 b | 4.07 ± 0.17 b | 8.38 ± 0.15 b | 9.27 ± 0.11 c | 4.65 ± 0.14 a | 4.62 ± 0.04 c | 44.88 ± 1.76 a | 57.27 ± 1.36 c |
| NT + BRZ | 2.88 ± 0.09 c | 10.85 ± 0.10 c | 3.77 ± 0.14 c | 7.08 ± 0.24 c | 8.05 ± 0.12 e | 4.08 ± 0.12 b | 3.97 ± 0.12 d | 43.75 ± 1.54 a | 55.72 ± 0.39 cd |
| HT + EBR | 3.20 ± 0.07 b | 12.32 ± 0.10 b | 4.10 ± 0.20 b | 8.23 ± 0.22 b | 9.74 ± 0.08 b | 4.62 ± 0.07 a | 5.12 ± 0.13 b | 41.50 ± 0.53 b | 70.73 ± 2.73 a |
| HT | 2.08 ± 0.08 d | 9.66 ± 0.26 d | 2.91 ± 0.09 d | 6.75 ± 0.17 c | 8.56 ± 0.12 d | 4.12 ± 0.09 b | 4.44 ± 0.21 c | 41.09 ± 0.47 b | 62.50 ± 0.57 b |
| HT + BRZ | 1.63 ± 0.01 e | 8.81 ± 0.09 e | 2.43 ± 0.05 e | 6.37 ± 0.13 d | 7.09 ± 0.21 f | 3.46 ± 0.13 c | 3.62 ± 0.08 e | 40.74 ± 0.33 b | 53.86 ± 1.05 d |
| BR, Brassinolide; Z, Zeatin; ZR, Zeatin riboside; iPA, N6-(Δ2-isopentenyl) adenosine riboside; aCTK, Active cytokinins (Z + ZR + iPA); GA1, Gibberellin A1; GA3, Gibberellin A3; GA4, Gibberellin A4; GA7, Gibberellin A7; aGA, Active gibberellins (GA1 + GA3 + GA4 + GA7); IAA, Indole-3-acetic acid; ABA, Abscisic acid. Values are Mean ± SD (n = 3). Different lowercase letters in the same column indicate significant differences between the treatments (P < 0.05). | |||||||||
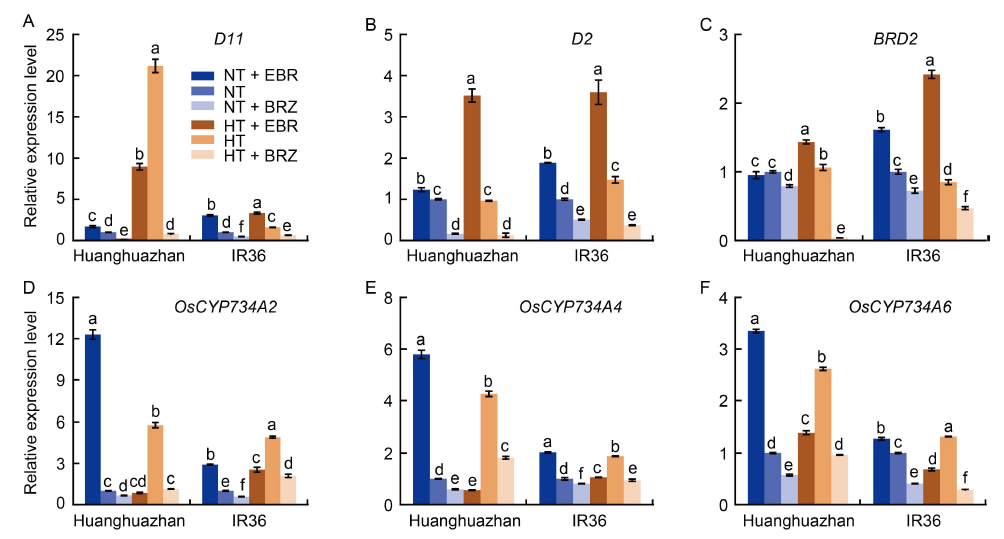
Fig. 2. Effects of exogenous 24-epibrassinolide (EBR) or brassinazole (BRZ) treatments on brassinolide biosynthesis (A?C) and catabolism (D?F) genes under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions. Transcript levels under NT conditions were set to 1.0. The samples were collected from the young panicles of rice on the 7th day of treatment. The qRT-PCR experiments had three biological replicates with OsUBQ as an internal reference gene. Values are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant differences between the treatments (P < 0.05).
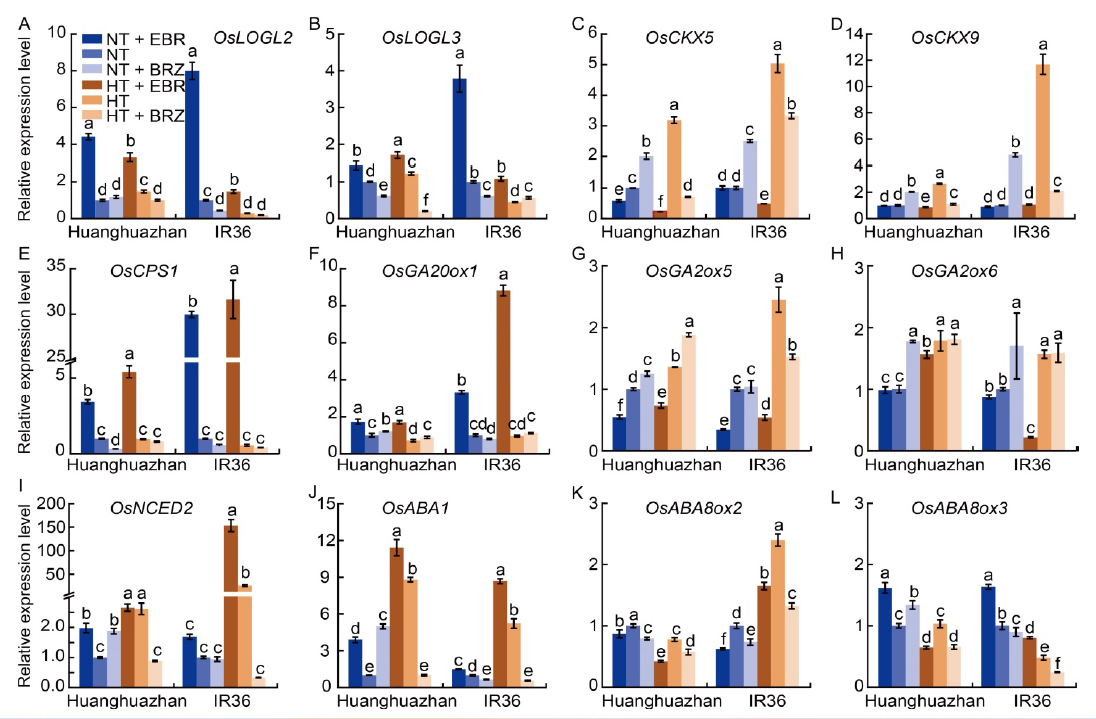
Fig. 3. Effects of exogenous 24-epibrassinolide (EBR) or brassinazole (BRZ) treatments on transcript levels of cytokinin biosynthesis and catabolism (A?D), gibberellin biosynthesis and catabolism (E?H), and abscisic acid biosynthesis and catabolism (I?L) genes under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions. Transcript levels under NT conditions were set to 1.0. The samples were collected from the young panicles of rice on the 7th day of treatment. The qRT-PCR experiments had three biological replicates with OsUBQ as an internal reference gene. Values are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant differences between the treatments (P < 0.05).
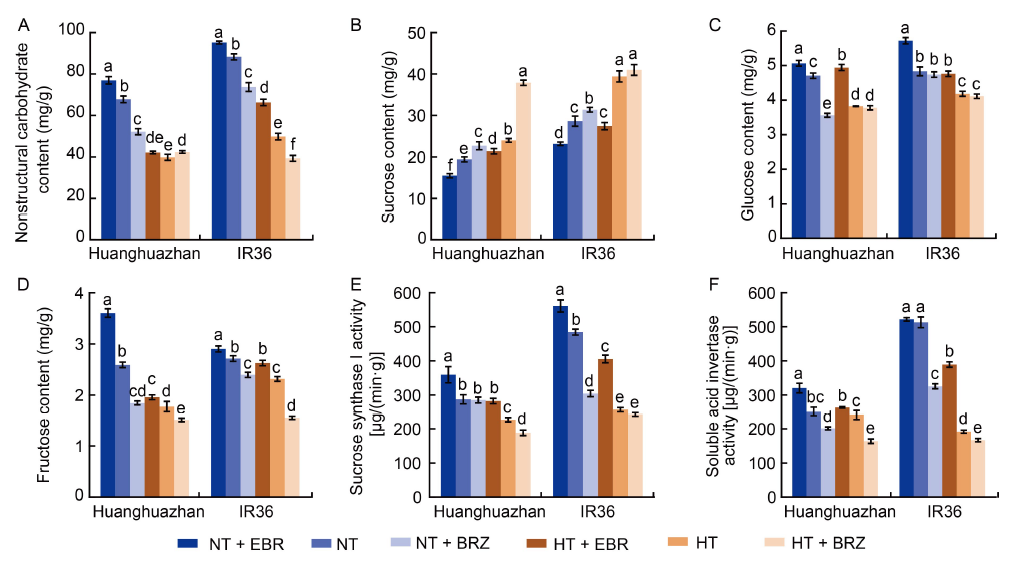
Fig. 4. Effects of exogenous 24-epibrassinolide (EBR) or brassinazole (BRZ) treatments on contents of nonstructural carbohydrate, sucrose, glucose, and fructose as well as activities of sucrose synthase I and soluble acid invertase in young panicles of rice under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions. A?F, Nonstructural carbohydrate content (A), sucrose content (B), glucose content (C), fructose content (D), sucrose synthase I activity (E) and soluble acid invertase activity (F) under different treatments. Values are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant differences between the treatments (P < 0.05).
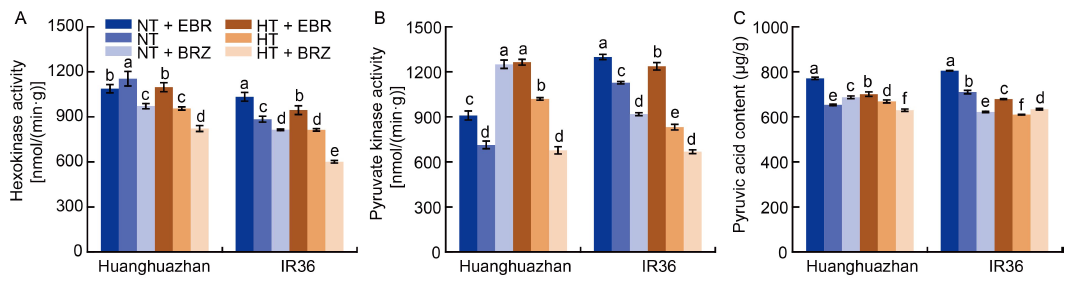
Fig. 5. Effects of exogenous 24-epibrassinolide (EBR) or brassinazole (BRZ) treatments on hexokinase and pyruvate kinase activities and pyruvic acid contents in young panicles of rice under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions. A?C, Hexokinase activity (A), pyruvate kinase activity (B) and pyruvic acid content (C) under different treatments. Values are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant differences between the treatments (P < 0.05).
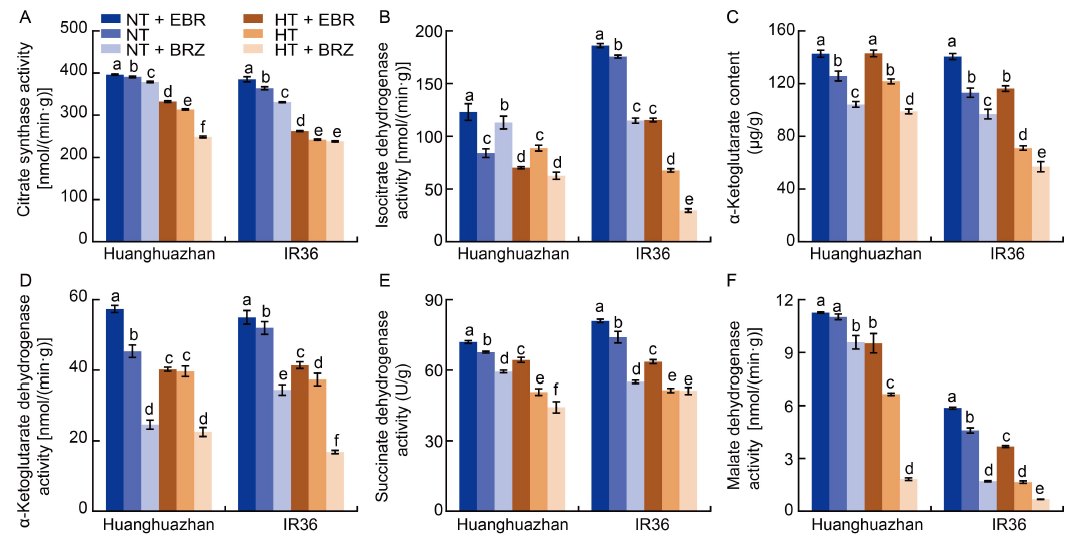
Fig. 6. Effects of exogenous 24-epibrassinolide (EBR) or brassinazole (BRZ) treatments on activities of key enzymes in tricarboxylic acidcycle in young panicles of rice under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions. A?F, Citrate synthase activity (A), isocitrate dehydrogenase activity (B), α-ketoglutarate content (C), α-ketoglutarate dehydrogenase activity (D), succinate dehydrogenase activity (E), and malate dehydrogenase activity (F) under different treatments. Values are Mean ± SD (n = 3). Different lowercase letters above the bars indicate significant differences between the treatments (P < 0.05).
| Cultivar | Treatment | H2O2 content (μmol/g) | MDA content (nmol/g) | SOD activity (U/g) | CAT activity [μmol/(min∙g)] | POD activity [ΔOD470/(min∙g)] |
| Huanghuazhan | NT + EBR | 5.16 ± 0.02 e | 9.36 ± 0.46 c | 947.56 ± 27.67 c | 194.89 ± 4.31 b | 1 758.83 ± 58.23 c |
| NT | 6.14 ± 0.03 d | 10.87 ± 0.23 b | 901.57 ± 20.82 c | 133.61 ± 2.08 c | 1 530.33 ± 22.06 d | |
| NT + BRZ | 8.35 ± 0.17 b | 11.20 ± 0.47 b | 913.96 ± 45.91 c | 384.93 ± 3.22 a | 1 720.67 ± 32.59 c | |
| HT + EBR | 6.32 ± 0.18 d | 10.73 ± 0.23 b | 1 184.12 ± 28.59 a | 136.00 ± 3.68 c | 2 085.67 ± 42.03 a | |
| HT | 7.34 ± 0.09 c | 11.06 ± 0.46 b | 1 114.53 ± 44.90 b | 135.02 ± 5.20 c | 2 034.50 ± 29.87 a | |
| HT + BRZ | 9.92 ± 0.22 a | 13.07 ± 0.54 a | 788.91 ± 24.60 d | 81.81 ± 3.54 d | 1 859.83 ± 43.28 b | |
| IR36 | NT + EBR | 6.24 ± 0.08 e | 13.17 ± 0.09 d | 956.59 ± 17.20 a | 427.76 ± 6.18 a | 2 025.50 ± 25.87 a |
| NT | 6.43 ± 0.11 d | 13.55 ± 0.32 d | 952.88 ± 16.06 a | 409.49 ± 4.25 b | 1 879.33 ± 29.17 b | |
| NT + BRZ | 7.90 ± 0.04 b | 14.08 ± 0.16 c | 747.56 ± 24.76 c | 274.87 ± 4.00 d | 1 663.83 ± 22.50 c | |
| HT + EBR | 6.36 ± 0.09 de | 13.60 ± 0.15 d | 852.33 ± 31.69 b | 312.02 ± 5.22 c | 1 841.67 ± 29.03 b | |
| HT | 7.42 ± 0.15 c | 15.51 ± 0.46 b | 767.49 ± 19.65 c | 184.90 ± 2.83 e | 1 497.83 ± 17.96 d | |
| HT + BRZ | 9.39 ± 0.06 a | 16.93 ± 0.19 a | 478.23 ± 30.11 d | 109.53 ± 4.87 f | 1 335.00 ± 39.85 c | |
| H2O2, Hydrogen peroxide; MDA, Malondialdehyde; SOD, Superoxide dismutase; CAT, Catalase; POD, Peroxidase. Values are Mean ± SD (n = 3). Different lowercase letters in the same column indicate significant differences between the treatments (P < 0.05). | ||||||
Table 3. Effects of exogenous 24-epibrassinolide (EBR) and brassinazole (BRZ) treatments on antioxidant ability in rice panicles under normal temperature (NT, 33 oC) and high temperature (HT, 40 oC) conditions.
| Cultivar | Treatment | H2O2 content (μmol/g) | MDA content (nmol/g) | SOD activity (U/g) | CAT activity [μmol/(min∙g)] | POD activity [ΔOD470/(min∙g)] |
| Huanghuazhan | NT + EBR | 5.16 ± 0.02 e | 9.36 ± 0.46 c | 947.56 ± 27.67 c | 194.89 ± 4.31 b | 1 758.83 ± 58.23 c |
| NT | 6.14 ± 0.03 d | 10.87 ± 0.23 b | 901.57 ± 20.82 c | 133.61 ± 2.08 c | 1 530.33 ± 22.06 d | |
| NT + BRZ | 8.35 ± 0.17 b | 11.20 ± 0.47 b | 913.96 ± 45.91 c | 384.93 ± 3.22 a | 1 720.67 ± 32.59 c | |
| HT + EBR | 6.32 ± 0.18 d | 10.73 ± 0.23 b | 1 184.12 ± 28.59 a | 136.00 ± 3.68 c | 2 085.67 ± 42.03 a | |
| HT | 7.34 ± 0.09 c | 11.06 ± 0.46 b | 1 114.53 ± 44.90 b | 135.02 ± 5.20 c | 2 034.50 ± 29.87 a | |
| HT + BRZ | 9.92 ± 0.22 a | 13.07 ± 0.54 a | 788.91 ± 24.60 d | 81.81 ± 3.54 d | 1 859.83 ± 43.28 b | |
| IR36 | NT + EBR | 6.24 ± 0.08 e | 13.17 ± 0.09 d | 956.59 ± 17.20 a | 427.76 ± 6.18 a | 2 025.50 ± 25.87 a |
| NT | 6.43 ± 0.11 d | 13.55 ± 0.32 d | 952.88 ± 16.06 a | 409.49 ± 4.25 b | 1 879.33 ± 29.17 b | |
| NT + BRZ | 7.90 ± 0.04 b | 14.08 ± 0.16 c | 747.56 ± 24.76 c | 274.87 ± 4.00 d | 1 663.83 ± 22.50 c | |
| HT + EBR | 6.36 ± 0.09 de | 13.60 ± 0.15 d | 852.33 ± 31.69 b | 312.02 ± 5.22 c | 1 841.67 ± 29.03 b | |
| HT | 7.42 ± 0.15 c | 15.51 ± 0.46 b | 767.49 ± 19.65 c | 184.90 ± 2.83 e | 1 497.83 ± 17.96 d | |
| HT + BRZ | 9.39 ± 0.06 a | 16.93 ± 0.19 a | 478.23 ± 30.11 d | 109.53 ± 4.87 f | 1 335.00 ± 39.85 c | |
| H2O2, Hydrogen peroxide; MDA, Malondialdehyde; SOD, Superoxide dismutase; CAT, Catalase; POD, Peroxidase. Values are Mean ± SD (n = 3). Different lowercase letters in the same column indicate significant differences between the treatments (P < 0.05). | ||||||
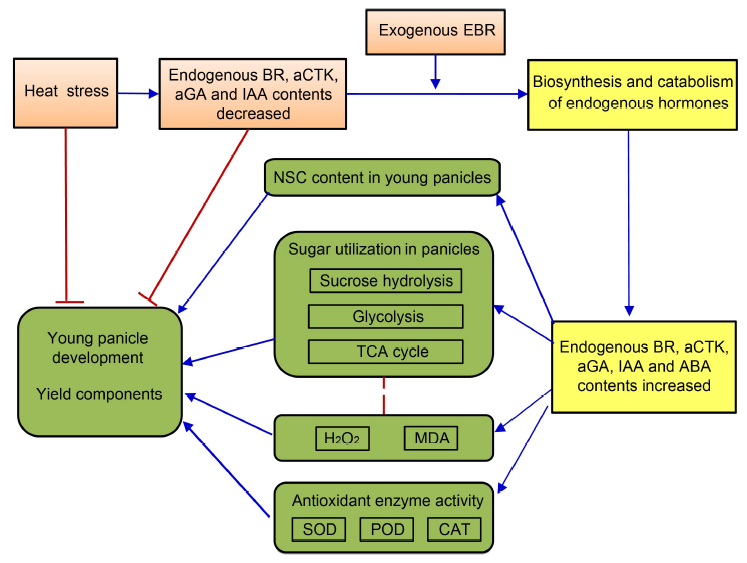
Fig. 7. Descriptive model of brassinosteroids mediate endogenous phytohormone metabolism to alleviate high temperature injury at panicle initiation stage in rice. High temperature decreases endogenous BR, aCTK, aGA and IAA contents, inhibits the activities of crucial enzymes involved in sucrose hydrolysis, glycolysis and the TCA cycle, and reduces the nonstructural carbohydrate content in young panicles of rice. In addition, high temperature decreases the activities of antioxidant enzymes and increases H2O2 and MDA contents, and ultimately inhibits young panicle development and reduces rice yield. Thus, a relationship between BR-regulated endogenous hormone metabolism and young panicle development is inferred. The blue ‘→’ indicates induction, and red ‘?’ indicates inhibition.BR, Brassinolide; EBR, 24-epibrassinolide; aCTK, Active cytokinins; aGA, Active gibberellins; IAA, Indole-3-acetic acid; ABA, Abscisic acid; NSC, Nonstructural carbohydrate; TCA, Tricarboxylic acid cycle; MDA, Malondialdehyde; SOD, Superoxide dismutase; POD, Peroxidase; CAT, Catalase.
| [1] | Aebi H. 1984. Catalase in vitro. Methods Enzymol, 105: 121-126. |
| [2] | Ali A, Xu P Z, Riaz A, Wu X J. 2019. Current advances in molecular mechanisms and physiological basis of panicle degeneration in rice. Int J Mol Sci, 20(7): 1613. |
| [3] |
Altmann T. 1998. Recent advances in brassinosteroid molecular genetics. Curr Opin Plant Biol, 1(5): 378-383.
PMID |
| [4] | Anuradha S, Rao S. 2003. Application of brassinosteroids to rice seeds (Oryza sativa L.) reduced the impact of salt stress on growth, prevented photosynthetic pigment loss and increased nitrate reductase activity. Plant Growth Regul, 40(1): 29-32. |
| [5] |
Anwar A, Liu Y M, Dong R R, Bai L Q, Yu X C, Li Y S. 2018. The physiological and molecular mechanism of brassinosteroid in response to stress: A review. Biol Res, 51(1): 46.
PMID |
| [6] | Arshad M S, Farooq M, Asch F, Krishna J S V, Prasad P V V, Siddique K H M. 2017. Thermal stress impacts reproductive development and grain yield in rice. Plant Physiol Biochem, 115: 57-72. |
| [7] |
Ashikari M, Sakakibara H, Lin S Y, Yamamoto T, Takashi T, Nishimura A, Angeles E R, Qian Q, Kitano H, Matsuoka M. 2005. Cytokinin oxidase regulates rice grain production. Science, 309: 741-745.
PMID |
| [8] | Bai M Y, Shang J X, Oh E, Fan M, Bai Y, Zentella R, Sun T P, Wang Z Y. 2012. Brassinosteroid, gibberellin and phytochrome impinge on a common transcription module in Arabidopsis. Nat Cell Biol, 14(8): 810-817. |
| [9] | Bajguz A, Hayat S. 2009. Effects of brassinosteroids on the plant responses to environmental stresses. Plant Physiol Biochem, 47(1): 1-8. |
| [10] |
Bolouri-Moghaddam M R, le Roy K, Xiang L, Rolland F, van den Ende W. 2010. Sugar signalling and antioxidant network connections in plant cells. FEBS J, 277(9): 2022-2037.
PMID |
| [11] |
Bouquin T, Meier C, Foster R, Nielsen M E, Mundy J. 2001. Control of specific gene expression by gibberellin and brassinosteroid. Plant Physiol, 127(2): 450-458.
PMID |
| [12] | Cai S L, Jiang G B, Ye N H, Chu Z Z, Xu X Z, Zhang J H, Zhu G H. 2015. A key ABA catabolic gene, OsABA8ox3, is involved in drought stress resistance in rice. PLoS One, 10(2): e0116646. |
| [13] | Cao Y Y, Zhao H. 2008. Protective roles of brassinolide on rice seedlings under high temperature stress. Rice Sci, 15(1): 63-68. |
| [14] |
Chen M, Lu Y, Ma Q, Guo L, Feng Y Q. 2009. Boronate affinity monolith for highly selective enrichment of glycopeptides and glycoproteins. Analyst, 134(10): 2158-2164.
PMID |
| [15] | Chen Y H, Wang Y L, Zhu D F, Shi Q H, Chen H Z, Xiang J, Zhang Y K, Zhang Y H. 2019. Mechanism of exogenous brassinolide in alleviating high temperature injury at panicle initiation stage in rice. Chin J Rice Sci, 33(5): 457-466. (in Chinese with English abstract) |
| [16] | Chen Y H, Chen H Z, Xiang J, Zhang Y K, Wang Z G, Zhu D F, Wang J K, Zhang Y P, Wang Y L. 2021. Rice spikelet formation inhibition caused by decreased sugar utilization under high temperature is associated with brassinolide decomposition. Environ Exp Bot, 190: 104585. |
| [17] |
Czechowski T, Bari R P, Stitt M, Scheible W R, Udvardi M K. 2004. Real-time RT-PCR profiling of over 1400 Arabidopsis transcription factors: Unprecedented sensitivity reveals novel root- and shoot-specific genes. Plant J, 38(2): 366-379.
PMID |
| [18] |
de Vleesschauwer D, van Buyten E, Satoh K, Balidion J, Mauleon R, Choi I R, Vera-Cruz C, Kikuchi S, Höfte M. 2012. Brassinosteroids antagonize gibberellin- and salicylate-mediated root immunity in rice. Plant Physiol, 158(4): 1833-1846.
PMID |
| [19] |
Ding J, Mao L J, Yuan B F, Feng Y Q. 2013. A selective pretreatment method for determination of endogenous active brassinosteroids in plant tissues: Double layered solid phase extraction combined with boronate affinity polymer monolith microextraction. Plant Methods, 9: 13.
PMID |
| [20] | Divi U K, Krishna P. 2009. Brassinosteroid: A biotechnological target for enhancing crop yield and stress tolerance. New Biotech, 26(3/4): 131-136. |
| [21] | DuBois M, Gilles K A, Hamilton J K, Rebers P A, Smith F. 1956. Colorimetric method for determination of sugars and related substances. Anal Chem, 28(3): 350-356. |
| [22] | Fageria N K. 2007. Yield physiology of rice. J Plant Nutr, 30(6): 843-879. |
| [23] |
Gao J, Chen H, Yang H F, He Y, Tian Z H, Li J X. 2018. A brassinosteroid responsive miRNA-target module regulates gibberellin biosynthesis and plant development. New Phytol, 220(2): 488-501.
PMID |
| [24] |
Giannopolitis C N, Ries S K. 1977. Superoxide dismutase: I. Occurrence in higher plants. Plant Physiol, 59(2): 309-314.
PMID |
| [25] | Ha Y M, Shang Y, Yang D M, Nam K H. 2018. Brassinosteroid reduces ABA accumulation leading to the inhibition of ABA- induced stomatal closure. Biochem Biophys Res Commun, 504(1): 143-148. |
| [26] |
Heath R L, Packer L. 1968. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys, 125(1): 189-198.
PMID |
| [27] |
Houba-Hérin N, Pethe C, d’Alayer J, Laloue M. 1999. Cytokinin oxidase from Zea mays: Purification, cDNA cloning and expression in moss protoplasts. Plant J, 17(6): 615-626.
PMID |
| [28] |
Hu Y X, Bao F, Li J Y. 2000. Promotive effect of brassinosteroids on cell division involves a distinct CycD3-induction pathway in Arabidopsis. Plant J, 24(5): 693-701.
PMID |
| [29] | Jiang N, Yu P H, Fu W M, Li G Y, Feng B H, Chen T T, Li H B, Tao L X, Fu G F. 2020. Acid invertase confers heat tolerance in rice plants by maintaining energy homoeostasis of spikelets. Plant Cell Environ, 43(5): 1273-1287. |
| [30] |
Kang J M, Turano F J. 2003. The putative glutamate receptor 1.1 (AtGLR1.1) functions as a regulator of carbon and nitrogen metabolism in Arabidopsis thaliana. Proc Natl Acad Sci USA, 100(11): 6872-6877.
PMID |
| [31] | Kang Y Y, Yang X, Guo S R, Zhang Y Y. 2011. Effects of 24- epibrassinolide on carbohydrate metabolism and enhancement of tolerance to root-zone hypoxia in cucumber (Cucumis sativus L.). Sci Agric Sin, 44(12): 2495-2503. (in Chinese with English abstract) |
| [32] | Kato K, Suzuki Y, Hosaka Y, Takahashi R, Kodama I, Sato K, Kawamoto T, Kumamaru T, Fujita N. 2019. Effect of high temperature on starch biosynthetic enzymes and starch structure in japonica rice cultivar ‘Akitakomachi’ (Oryza sativa L.) endosperm and palatability of cooked rice. J Cereal Sci, 87: 209-214. |
| [33] |
Li G Y, Zhang C X, Zhang G H, Fu W M, Feng B H, Chen T T, Peng S B, Tao L X, Fu G F. 2020. Abscisic acid negatively modulates heat tolerance in rolled leaf rice by increasing leaf temperature and regulating energy homeostasis. Rice, 13(1): 18.
PMID |
| [34] |
Li Q Q, Xu F, Chen Z, Teng Z F, Sun K, Li X C, Yu J Y, Zhang G X, Liang Y, Huang X H, Du L, Qian Y W, Wang Y C, Chu C C, Tang J Y. 2021. Synergistic interplay of ABA and BR signal in regulating plant growth and adaptation. Nat Plants, 7(8): 1108-1118.
PMID |
| [35] | Liu D, Guo G H, Liu L, Zou C L, Wu P R, Yang F F, Wang Y B, Li C F. 2018. Effects of exogenous application of brassinolide on sugar beet growth under alkali stress. Acta Agric Bor-Occid Sin, 27(10): 1461-1469. (in Chinese with English abstract) |
| [36] | Liu X L, Zhang H, Jin Y Y, Wang M M, Yang H Y, Ma H Y, Jiang C J, Liang Z W. 2019. Abscisic acid primes rice seedlings for enhanced tolerance to alkaline stress by upregulating antioxidant defense and stress tolerance-related genes. Plant Soil, 438(1/2): 39-55. |
| [37] | Lu Y, Yamaguchi J, Sato T. 2015. Integration of C/N-nutrient and multiple environmental signals into the ABA signaling cascade. Plant Signal Behav, 10(12): e1048940. |
| [38] |
Lv J H, Dong T Y, Zhang Y P, Ku Y, Zheng T, Jia H F, Fang J G. 2022. Metabolomic profiling of brassinolide and abscisic acid in response to high-temperature stress. Plant Cell Rep, 41(4): 935-946.
PMID |
| [39] | Maehly A, Chance B. 1954. Catalases and peroxidases, part II. Methods Biochem Anal, 1: 357-424. |
| [40] | Matusmoto T, Yamada K, Yoshizawa Y, Oh K. 2016. Comparison of effect of brassinosteroid and gibberellin biosynthesis inhibitors on growth of rice seedlings. Rice Sci, 23(1): 51-55. |
| [41] | McCready R M, Guggolz J, Silviera V, Owens H S. 1950. Determination of starch and amylose in vegetables. Anal Chem, 22(9): 1156-1158. |
| [42] |
Miyawaki K, Matsumoto-Kitano M, Kakimoto T. 2004. Expression of cytokinin biosynthetic isopentenyltransferase genes in Arabidopsis: Tissue specificity and regulation by auxin, cytokinin, and nitrate. Plant J, 37(1): 128-138.
PMID |
| [43] |
Nemhauser J L, Hong F X, Chory J. 2006. Different plant hormones regulate similar processes through largely nonoverlapping transcriptional responses. Cell, 126(3): 467-475.
PMID |
| [44] | Pantoja-Benavides A D, Garces-Varon G, Restrepo-Díaz H. 2021. Foliar growth regulator sprays induced tolerance to combined heat stress by enhancing physiological and biochemical responses in rice. Front Plant Sci, 12: 702892. |
| [45] | Patel R, Mohapatra P K. 1992. Regulation of spikelet development in rice by hormones. J Exp Bot, 43(2): 257-262. |
| [46] |
Rao M V, Lee H, Creelman R A, Mullet J E, Davis K R. 2000. Jasmonic acid signaling modulates ozone-induced hypersensitive cell death. Plant Cell, 12(9): 1633-1646.
PMID |
| [47] |
Reid E E, Thompson P, Lyttle C R, Dennis D T. 1977. Pyruvate dehydrogenase complex from higher plant mitochondria and proplastids. Plant Physiol, 59(5): 842-848.
PMID |
| [48] | Sánchez B, Rasmussen A, Porter J R. 2014. Temperatures and the growth and development of maize and rice: A review. Glob Chang Biol, 20(2): 408-417. |
| [49] | Setsungnern A, Muñoz P, Pérez-Llorca M, Müller M, Thiravetyan P, Munné-Bosch S. 2020. A defect in BRI1-EMS-SUPPRESSOR 1 (bes1)-mediated brassinosteroid signaling increases photoinhibition and photo-oxidative stress during heat stress in Arabidopsis. Plant Sci, 296: 110470. |
| [50] | Sharma M, Mahajan P, Singh H P, Batish D R, Kohli R K. 2019. 24-Epibrassinolide pre-treatment reduces alkaline-induced oxidative stress in red rice seedlings. Environ Sci Pollut Res Int, 26(22): 23192-23197. |
| [51] | Shinohara T, Leskovar D I. 2014. Effects of ABA, antitranspirants, heat and drought stress on plant growth, physiology and water status of artichoke transplants. Sci Hortic, 165: 225-234. |
| [52] | Skazhennik M A, Vorob’yov N V, Sheudzhen A K, Kovalyov V S. 2015. Causes of increased panicle spikelet sterility in rice. Russ Agric Sci, 41(5): 309-310. |
| [53] | Tang R S, Zheng J C, Jin Z Q, Zhang D D, Huang Y H, Chen L G. 2008. Possible correlation between high temperature-induced floret sterility and endogenous levels of IAA, GAs and ABA in rice (Oryza sativa L.). Plant Growth Regul, 54(1): 37-43. |
| [54] |
Tang S, Zhang H X, Li L, Liu X, Chen L, Chen W Z, Ding Y F. 2018. Exogenous spermidine enhances the photosynthetic and antioxidant capacity of rice under heat stress during early grain-filling period. Funct Plant Biol, 45(9): 911-921.
PMID |
| [55] |
Toh S, Imamura A, Watanabe A, Nakabayashi K, Okamoto M, Jikumaru Y, Hanada A, Aso Y, Ishiyama K, Tamura N, Iuchi S, Kobayashi M, Yamaguchi S, Kamiya Y, Nambara E, Kawakami N. 2008. High temperature-induced abscisic acid biosynthesis and its role in the inhibition of gibberellin action in Arabidopsis seeds. Plant Physiol, 146(3): 1368-1385.
PMID |
| [56] | Tong H N, Xiao Y H, Liu D P, Gao S P, Liu L C, Yin Y H, Jin Y, Qian Q, Chu C C. 2014. Brassinosteroid regulates cell elongation by modulating gibberellin metabolism in rice. Plant Cell, 26(11): 4376-4393. |
| [57] |
Wang H J, Tang J, Liu J, Hu J, Liu J J, Chen Y X, Cai Z Y, Wang X L. 2018. Abscisic acid signaling inhibits brassinosteroid signaling through dampening the dephosphorylation of BIN2 by ABI1 and ABI2. Mol Plant, 11(2): 315-325.
PMID |
| [58] | Wang S Q, Zhao H H, Zhao L M, Gu C M, Na Y G, Xie B S, Cheng S H, Pan G J. 2020. Application of brassinolide alleviates cold stress at the booting stage of rice. J Integr Agric, 19(4): 975-987. |
| [59] | Wang W T, Cui W P, Xu K, Gao H, Wei H Y, Zhang H C. 2021. Effects of early- and late-sowing on starch accumulation and associated enzyme activities during grain filling stage in rice. Rice Sci, 28(2): 191-199. |
| [60] | Wang X, Liu D, Chen J T, Liu X Y, Zhang H, Wang Y B, Li C F. 2021. Effects of exogenous BR on endogenous hormone and protective enzyme activities in sugar beet under saline-alkali stress. J Northwest A & F Univ: Nat Sci, 49(7): 20-30. (in Chinese with English abstract) |
| [61] | Wang Y L, Zhang Y K, Zhang Q, Cui Y T, Xiang J, Chen H Z, Hu G H, Chen Y H, Wang X D, Zhu D F, Zhang Y P. 2019. Comparative transcriptome analysis of panicle development under heat stress in two rice (Oryza sativa L.) cultivars differing in heat tolerance. PeerJ, 7: e7595. |
| [62] | Wang Y L, Zhang Y K, Shi Q H, Chen H Z, Xiang J, Hu G H, Chen Y H, Wang X D, Wang J K, Yi Z H, Zhu D F, Zhang Y P. 2020. Decrement of sugar consumption in rice young panicle under high temperature aggravates spikelet number reduction. Rice Sci, 27(1): 44-55. |
| [63] | Wang Z. 2000. Plant Physiology. Beijing, China:China Agricultural Press: 303-365. (in Chinese) |
| [64] |
Weiss D, Ori N. 2007. Mechanisms of cross talk between gibberellin and other hormones. Plant Physiol, 144(3): 1240-1246.
PMID |
| [65] |
Werner T, Schmülling T. 2009. Cytokinin action in plant development. Curr Opin Plant Biol, 12(5): 527-538.
PMID |
| [66] |
Wu C, Cui K H, Wang W C, Li Q, Fahad S, Hu Q Q, Huang J L, Nie L X, Peng S B. 2016. Heat-induced phytohormone changes are associated with disrupted early reproductive development and reduced yield in rice. Sci Rep, 6: 34978.
PMID |
| [67] |
Wu C, Cui K H, Wang W C, Li Q, Fahad S, Hu Q Q, Huang J L, Nie L X, Mohapatra P K, Peng S B. 2017. Heat-induced cytokinin transportation and degradation are associated with reduced panicle cytokinin expression and fewer spikelets per panicle in rice. Front Plant Sci, 8: 371.
PMID |
| [68] | Wu C, Tang S, Li G H, Wang S H, Fahad S, Ding Y F. 2019. Roles of phytohormone changes in the grain yield of rice plants exposed to heat: A review. PeerJ, 7: e7792. |
| [69] | Xu Y F, Chu C C, Yao S G. 2021. The impact of high-temperature stress on rice: Challenges and solutions. Crop J, 9(5): 963-976. |
| [70] | Yan L Y, Zhang H J, Zheng Y Q, Cong Y Q, Liu C T, Fan F, Zheng C, Yuan G L, Pan G, Yuan D Y, Duan M J. 2021. Transcription factor OsMADS25 improves rice tolerance to cold stress. Hereditas, 43(11): 1078-1087. (in Chinese with English abstract) |
| [71] |
Yang J C, Zhang J H, Wang Z Q, Zhu Q S, Wang W. 2001. Hormonal changes in the grains of rice subjected to water stress during grain filling. Plant Physiol, 127(1): 315-323.
PMID |
| [72] | Yang Y Y, Chen X, Chen Q Z, Lu F, Xu C, Yang H T, Su P P, Liu X L. 2021. Priming effects of abscisic acid on high temperature stress tolerance in rice at seed germination stage. Acta Agric Boreal-Sin, 36(3): 185-194. (in Chinese with English abstract) |
| [73] |
Yu J Q, Huang L F, Hu W H, Zhou Y H, Mao W H, Ye S F, Nogués S. 2004. A role for brassinosteroids in the regulation of photosynthesis in Cucumis sativus. J Exp Bot, 55: 1135-1143.
PMID |
| [74] | Yuan G F, Jia C G, Li Z, Sun B, Zhang L P, Liu N, Wang Q M. 2010. Effect of brassinosteroids on drought resistance and abscisic acid concentration in tomato under water stress. Sci Hortic, 126(2): 103-108. |
| [75] | Yuldashev R, Avalbaev A, Bezrukova M, Vysotskaya L, Khripach V, Shakirova F. 2012. Cytokinin oxidase is involved in the regulation of cytokinin content by 24-epibrassinolide in wheat seedlings. Plant Physiol Biochem, 55: 1-6. |
| [76] |
Zhang A Y, Zhang J, Zhang J H, Ye N H, Zhang H, Tan M P, Jiang M Y. 2011. Nitric oxide mediates brassinosteroid-induced ABA biosynthesis involved in oxidative stress tolerance in maize leaves. Plant Cell Physiol, 52(1): 181-192.
PMID |
| [77] | Zhang D P, Zhang M Y, Wang Y Z, Liang J S. 2021. RGB1 regulates rice panicle architecture and grain filling through monitoring cytokinin level in inflorescence meristem and grain abscisic acid level during filling stage. Rice Sci, 28(4): 317-321. |
| [78] |
Zhang S S, Cai Z Y, Wang X L. 2009. The primary signaling outputs of brassinosteroids are regulated by abscisic acid signaling. Proc Natl Acad Sci USA, 106(11): 4543-4548.
PMID |
| [79] | Zhang W Y, Sheng J Y, Xu Y J, Xiong F, Wu Y F, Wang W L, Wang Z Q, Yang J C, Zhang J H. 2019. Role of brassinosteroids in rice spikelet differentiation and degeneration under soil- drying during panicle development. BMC Plant Biol, 19(1): 409. |
| [80] | Zhang W Y, Sheng J Y, Fu L D, Xu Y J, Xiong F, Wu Y F, Wang W L, Wang Z Q, Zhang J H, Yang J C. 2020. Brassinosteroids mediate the effect of soil-drying during meiosis on spikelet degeneration in rice. Environ Exp Bot, 169: 103887. |
| [81] | Zhang Y J. 1977. Determination of glucose, fructose, sucrose and starch in fruit and vegatable with anthrone colorimetric method. Chin J Anal Chem, 5(3): 167-171. (in Chinese) |
| [82] |
Zhao C, Liu B, Piao S L, Wang X H, Lobell D B, Huang Y, Huang M T, Yao Y T, Bassu S, Ciais P, Durand J L, Elliott J, Ewert F, Janssens I A, Li T, Lin E D, Liu Q, Martre P, Müller C, Peng S S, Peñuelas J, Ruane A C, Wallach D, Wang T, Wu D H, Liu Z, Zhu Y, Zhu Z C, Asseng S. 2017. Temperature increase reduces global yields of major crops in four independent estimates. Proc Natl Acad Sci USA, 114(35): 9326-9331.
PMID |
| [83] | Zhao X S, Wang Q, Yan Q, Zhao Y, Wang F R, Dong J G. 2016. Function of brassinolide in the regulation of root development in rice. Chin J Cell Biol, 38(10): 1191-1198. (in Chinese with English abstract) |
| [84] | Zhu J K. 2002. Salt and drought stress signal transduction in plants. Annu Rev Plant Biol, 53: 247-273. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [13] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||