
Rice Science ›› 2016, Vol. 23 ›› Issue (5): 242-254.DOI: 10.1016/j.rsci.2016.08.002
• Orginal Article • Previous Articles Next Articles
Chandran Jisha Kolothodi, Thomas Puthur Jos( )
)
Received:2015-10-14
Accepted:2015-12-09
Online:2016-09-05
Published:2016-06-12
Chandran Jisha Kolothodi, Thomas Puthur Jos. Seed Priming with Beta-Amino Butyric Acid Improves Abiotic Stress Tolerance in Rice Seedlings[J]. Rice Science, 2016, 23(5): 242-254.
Add to citation manager EndNote|Ris|BibTeX
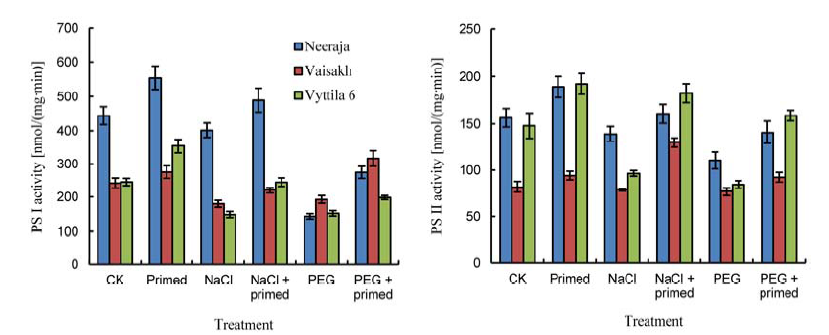
Fig. 1. Effects of beta-amino butyric acid priming on photosystem activities. CK, Control; PEG, PEG-6000. Bars represent SE of the mean values (n = 9).
| Trait | Variety | Control | Primed | NaCl | NaCl + primed | PEG-6000 | PEG-6000 + primed |
|---|---|---|---|---|---|---|---|
| Shoot length (cm) | Neeraja | 9.13 ± 0.32 b | 11.40 ± 0.51 a | 5.57 ± 0.34 c | 10.03 ± 0.54 ab | 4.53 ± 0.17 c | 5.17 ± 0.55 c |
| Vaisakh | 10.20 ± 0.43 b | 10.57 ± 0.42 b | 4.77 ± 0.12 d | 12.53 ± 0.33 a | 4.37 ± 0.12 d | 8.67 ± 0.36 c | |
| Vyttila 6 | 11.77 ± 0.25 a | 11.83 ± 0.19 a | 4.90 ± 0.13 d | 8.07 ± 0.34 bc | 8.07 ± 0.34 bc | 9.20 ± 0.48 b | |
| Fresh weight (g) | Neeraja | 0.0449 ± 0.0121 c | 0.0590 ± 0.0133 a | 0.0427 ± 0.0124 cd | 0.0536 ± 0.0120 b | 0.0394 ± 0.0103 d | 0.0446 ± 0.0104 c |
| Vaisakh | 0.0569 ± 0.0123 c | 0.0665 ± 0.0102 b | 0.0480 ± 0.0100 d | 0.0813 ± 0.0112 a | 0.0463 ± 0.0114 d | 0.0674 ± 0.0091 b | |
| Vyttila 6 | 0.0725 ± 0.0108 ab | 0.0839 ± 0.0112 a | 0.0505 ± 0.0991 d | 0.0586 ± 0.0105 cd | 0.0572 ± 0.0113 cd | 0.0663 ± 0.0112 bc | |
| Dry weight (g) | Neeraja | 0.0136 ± 0.0099 bc | 0.0156 ± 0.0104 a | 0.0126 ± 0.0114 c | 0.0163 ± 0.0121 a | 0.0130 ± 0.0114 bc | 0.0140 ± 0.0120 b |
| Vaisakh | 0.0131 ± 0.0113 c | 0.0155 ± 0.0114 a | 0.0116 ± 0.0132 d | 0.0150 ± 0.0131 a | 0.0116 ± 0.0112 d | 0.0139 ± 0.0109 b | |
| Vyttila 6 | 0.0131 ± 0.0105 b | 0.0146 ± 0.0103 a | 0.0115 ± 0.0106 d | 0.0129 ± 0.0134 b | 0.0120 ± 0.0142 cd | 0.0125 ± 0.0114 bc |
Table 1 Effects of beta-amino butyric acid seed priming on seedling growth parameters of Neeraja, Vaisakh and Vyttila 6.
| Trait | Variety | Control | Primed | NaCl | NaCl + primed | PEG-6000 | PEG-6000 + primed |
|---|---|---|---|---|---|---|---|
| Shoot length (cm) | Neeraja | 9.13 ± 0.32 b | 11.40 ± 0.51 a | 5.57 ± 0.34 c | 10.03 ± 0.54 ab | 4.53 ± 0.17 c | 5.17 ± 0.55 c |
| Vaisakh | 10.20 ± 0.43 b | 10.57 ± 0.42 b | 4.77 ± 0.12 d | 12.53 ± 0.33 a | 4.37 ± 0.12 d | 8.67 ± 0.36 c | |
| Vyttila 6 | 11.77 ± 0.25 a | 11.83 ± 0.19 a | 4.90 ± 0.13 d | 8.07 ± 0.34 bc | 8.07 ± 0.34 bc | 9.20 ± 0.48 b | |
| Fresh weight (g) | Neeraja | 0.0449 ± 0.0121 c | 0.0590 ± 0.0133 a | 0.0427 ± 0.0124 cd | 0.0536 ± 0.0120 b | 0.0394 ± 0.0103 d | 0.0446 ± 0.0104 c |
| Vaisakh | 0.0569 ± 0.0123 c | 0.0665 ± 0.0102 b | 0.0480 ± 0.0100 d | 0.0813 ± 0.0112 a | 0.0463 ± 0.0114 d | 0.0674 ± 0.0091 b | |
| Vyttila 6 | 0.0725 ± 0.0108 ab | 0.0839 ± 0.0112 a | 0.0505 ± 0.0991 d | 0.0586 ± 0.0105 cd | 0.0572 ± 0.0113 cd | 0.0663 ± 0.0112 bc | |
| Dry weight (g) | Neeraja | 0.0136 ± 0.0099 bc | 0.0156 ± 0.0104 a | 0.0126 ± 0.0114 c | 0.0163 ± 0.0121 a | 0.0130 ± 0.0114 bc | 0.0140 ± 0.0120 b |
| Vaisakh | 0.0131 ± 0.0113 c | 0.0155 ± 0.0114 a | 0.0116 ± 0.0132 d | 0.0150 ± 0.0131 a | 0.0116 ± 0.0112 d | 0.0139 ± 0.0109 b | |
| Vyttila 6 | 0.0131 ± 0.0105 b | 0.0146 ± 0.0103 a | 0.0115 ± 0.0106 d | 0.0129 ± 0.0134 b | 0.0120 ± 0.0142 cd | 0.0125 ± 0.0114 bc |
| Variety | Chlorophyll a | Chlorophyll b | ||||
|---|---|---|---|---|---|---|
| Control | NaCl | PEG-6000 | Control | NaCl | PEG-6000 | |
| Neeraja | 2.02 ± 0.16 b (1.58 ± 0.15 d) | 2.26 ± 0.16 a (1.52 ± 0.13 e) | 1.70 ± 0.11 c (1.15 ± 0.21 f) | 0.57 ± 0.05 b (0.40 ± 0.03 e) | 0.65 ± 0.04 a (0.43 ± 0.03 d) | 0.50 ± 0.04 c (0.30 ± 0.03 f) |
| Vaisakh | 2.82 ± 0.06 a (2.16 ± 0.05 d) | 2.58 ± 0.15 c (2.00 ± 0.14 e) | 2.76 ± 0.04 b (1.10 ± 0.06 f) | 0.70 ± 0.03 b (0.51 ± 0.01 d) | 0.59 ± 0.05 c (0.49 ± 0.04 e) | 0.72 ± 0.03 a (0.31 ± 0.02 f) |
| Vyttila 6 | 3.71 ± 0.16 c (2.84 ± 0.05 d) | 4.04 ± 0.06 a (2.53 ± 0.05 f) | 4.03 ± 0.13 b (2.74 ± 0.18 e) | 1.14 ± 0.05 c (0.83 ± 0.04 e) | 1.20 ± 0.04 b (0.77 ± 0.03 f) | 1.29 ± 0.06 a (0.89 ± 0.06 d) |
| Variety | Total chlorophyll | Carotenoid | ||||
| Control | NaCl | PEG-6000 | Control | NaCl | PEG-6000 | |
| Neeraja | 2.59 ± 0.16 b (1.97 ± 0.14 d) | 2.90 ± 0.15 a (1.95 ± 0.13 e) | 2.19 ± 0.15 c (1.45 ± 0.11 f) | 0.89 ± 0.02 b (0.65 ± 0.02 d) | 0.96 ± 0.05 a (0.65 ± 0.06 d) | 0.70 ± 0.03 c (0.46 ± 0.06 e) |
| Vaisakh | 3.51 ± 0.15 a (2.66 ± 0.16 d) | 3.16 ± 0.15 c (2.49 ± 0.11 e) | 3.48 ± 0.14 b (1.41 ± 0.10 f) | 1.29 ± 0.05 b (1.10 ± 0.04 d) | 1.23 ± 0.04 c (1.04 ± 0.05 e) | 1.30 ± 0.05 a (0.71 ± 0.06 f) |
| Vyttila 6 | 4.84 ± 0.12 c (3.67 ± 0.11 d) | 5.26 ± 0.17 b (3.30 ± 0.18 f) | 5.32 ± 0.15 a (3.62 ± 0.14 e) | 1.41 ± 0.04 c (1.17 ± 0.07 d) | 1.46 ± 0.04 b (0.99 ± 0.02 f) | 1.61 ± 0.06 a (1.01 ± 0.05 e) |
Table 2 Photosynthetic pigment content in seedling leaves under beta-amino butyric acid priming (values without parenthesis) and non-primed seeds (values in parenthesis).
| Variety | Chlorophyll a | Chlorophyll b | ||||
|---|---|---|---|---|---|---|
| Control | NaCl | PEG-6000 | Control | NaCl | PEG-6000 | |
| Neeraja | 2.02 ± 0.16 b (1.58 ± 0.15 d) | 2.26 ± 0.16 a (1.52 ± 0.13 e) | 1.70 ± 0.11 c (1.15 ± 0.21 f) | 0.57 ± 0.05 b (0.40 ± 0.03 e) | 0.65 ± 0.04 a (0.43 ± 0.03 d) | 0.50 ± 0.04 c (0.30 ± 0.03 f) |
| Vaisakh | 2.82 ± 0.06 a (2.16 ± 0.05 d) | 2.58 ± 0.15 c (2.00 ± 0.14 e) | 2.76 ± 0.04 b (1.10 ± 0.06 f) | 0.70 ± 0.03 b (0.51 ± 0.01 d) | 0.59 ± 0.05 c (0.49 ± 0.04 e) | 0.72 ± 0.03 a (0.31 ± 0.02 f) |
| Vyttila 6 | 3.71 ± 0.16 c (2.84 ± 0.05 d) | 4.04 ± 0.06 a (2.53 ± 0.05 f) | 4.03 ± 0.13 b (2.74 ± 0.18 e) | 1.14 ± 0.05 c (0.83 ± 0.04 e) | 1.20 ± 0.04 b (0.77 ± 0.03 f) | 1.29 ± 0.06 a (0.89 ± 0.06 d) |
| Variety | Total chlorophyll | Carotenoid | ||||
| Control | NaCl | PEG-6000 | Control | NaCl | PEG-6000 | |
| Neeraja | 2.59 ± 0.16 b (1.97 ± 0.14 d) | 2.90 ± 0.15 a (1.95 ± 0.13 e) | 2.19 ± 0.15 c (1.45 ± 0.11 f) | 0.89 ± 0.02 b (0.65 ± 0.02 d) | 0.96 ± 0.05 a (0.65 ± 0.06 d) | 0.70 ± 0.03 c (0.46 ± 0.06 e) |
| Vaisakh | 3.51 ± 0.15 a (2.66 ± 0.16 d) | 3.16 ± 0.15 c (2.49 ± 0.11 e) | 3.48 ± 0.14 b (1.41 ± 0.10 f) | 1.29 ± 0.05 b (1.10 ± 0.04 d) | 1.23 ± 0.04 c (1.04 ± 0.05 e) | 1.30 ± 0.05 a (0.71 ± 0.06 f) |
| Vyttila 6 | 4.84 ± 0.12 c (3.67 ± 0.11 d) | 5.26 ± 0.17 b (3.30 ± 0.18 f) | 5.32 ± 0.15 a (3.62 ± 0.14 e) | 1.41 ± 0.04 c (1.17 ± 0.07 d) | 1.46 ± 0.04 b (0.99 ± 0.02 f) | 1.61 ± 0.06 a (1.01 ± 0.05 e) |
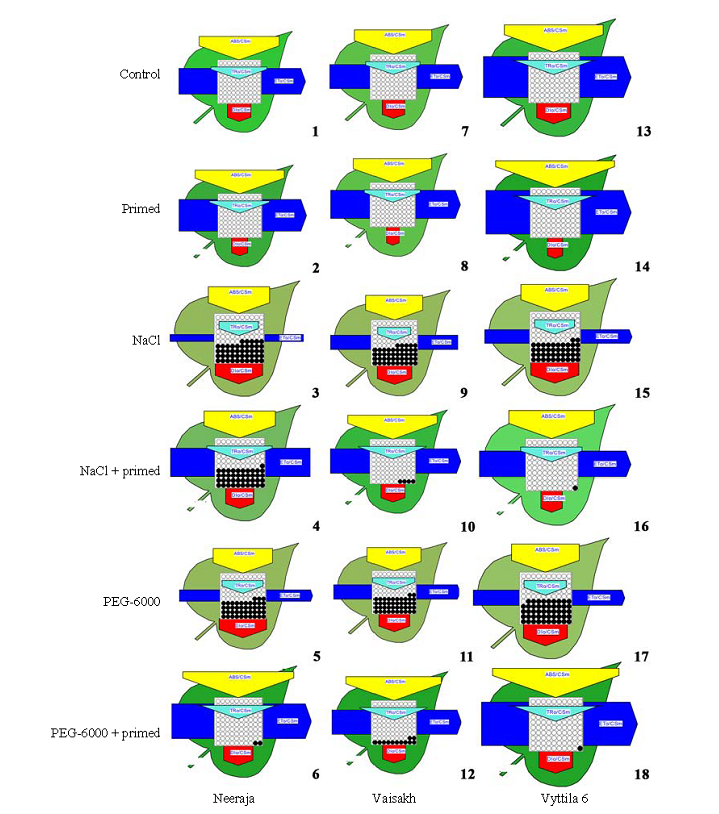
Fig. 2. Energy pipeline leaf model of phenomenological fluxes (per cross section, CS) in three rice varieties. 1-6 (Neeraja), 7-12 (Vaisakh) and 13-18 (Vyttila 6) subjected to different treatments (control, primed, NaCl, NaCl + primed, PEG-6000 and PEG-6000 + primed, respectively). The value of each parameter can be seen in relative changes in width of each arrow. Active reaction centres are shown as open circles and inactive reaction centres are shown as closed circles.
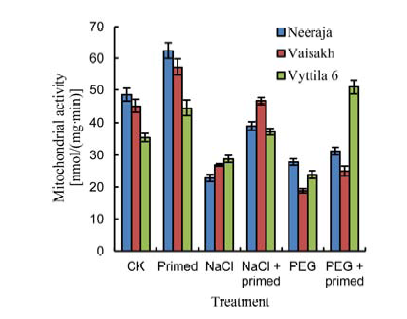
Fig. 3. Effects of beta-amino butyric acid priming on mitochondrial activity of three rice varieties. CK, Control; PEG, PEG-6000. Bars represent SE of the mean values (n = 9).
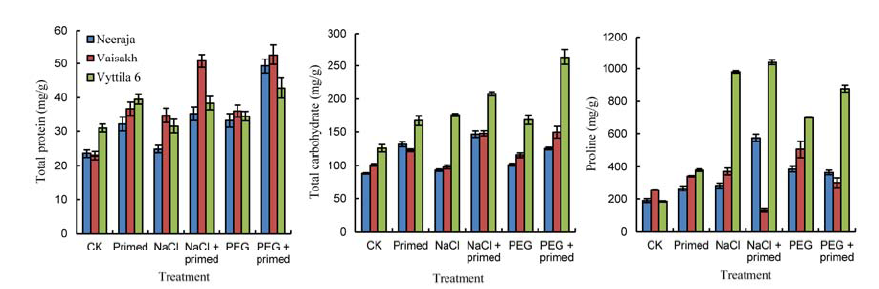
Fig. 4. Effects of beta-amino butyric acid priming on total protein, total carbohydrate and proline contents of three rice varieties. CK, Control; PEG, PEG-6000. Bars represent SE of the mean values (n = 9).
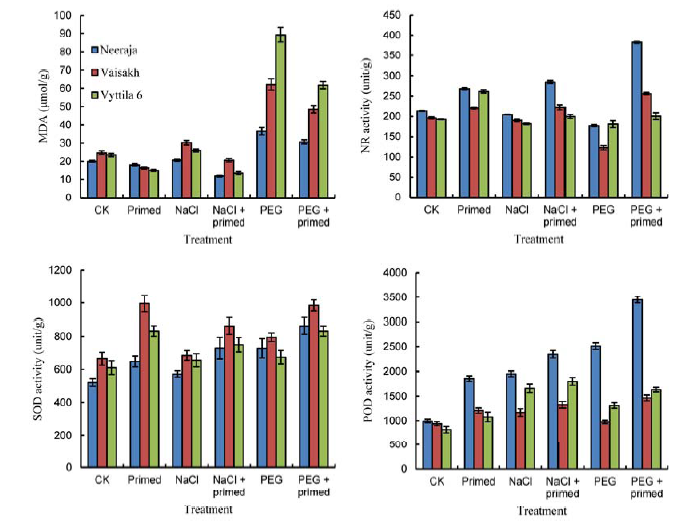
Fig. 5. Effects of beta-amino butyric acid priming on malondialdehyde (MDA) content, nitrate reductase (NR), superoxide dismutase (SOD) and guaiacol peroxidase (POD) activities of three rice varieties. CK, Control; PEG, PEG-6000. Bars represent SE of the mean values (n = 9).
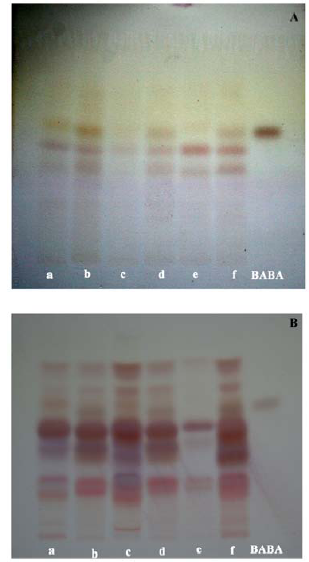
Fig. 6. Detection of beta-amino butyric acid (BABA) in seeds (A) and seedlings (B) of three rice varieties. a, Neeraja (CK); b, BABA primed Neeraja; c, Vaisakh (CK); d, BABA primed Vaisakh; e, Vyttila 6 (CK); f, BABA primed Vyttila 6.
| 1 | Aghbolaghi M A, Sedghi M.2014. The effect of osmo and hormone priming on germination and seed reserve utilization of millet seeds under drought stress. J Stress Physiol Biochem, 10(1): 214-221. |
| 2 | Arnon D I.1949. Copper enzymes in isolated chloroplasts polyphenoloxidase in Beta vulgaris.Plant Physiol, 24(1): 1-5. |
| 3 | Ashraf M Y, Azmi A R, Khan A H, Ala S A.1994. Effect of water stress on total phenols peroxidase activity and chlorophyll content in wheat (Triticum aestivum L.).Acta Physiol Plant, 16(3): 185-191. |
| 4 | Ashraf M, Bray C M.1993. DNA synthesis in osmoprimed leek (Allium porrum L.) seeds and evidence for repair and replication.Seed Sci Res, 3: 15-23. |
| 5 | Ashraf M, Foolad R M.2005. Pre-sowing seed treatment: A shotgun approach to improve germination, plant growth and crop yield under saline and non-saline conditions.Adv Agron, 88: 223-271. |
| 6 | Bagheri M Z.2014. The effect of maize priming on germination characteristics, catalase and peroxidase enzyme activity and total protein content under salt stress.Int J Biosci, 4(2): 104-112. |
| 7 | Bajehbaj A A.2010. The effects of NaCl priming on salt tolerance in sunflower germination and seedling grown under salinity conditions.Afr J Biotechnol, 9: 1764-1770. |
| 8 | Bates L S, Waldren R P, Teare I D.1973. Rapid determination of free proline for water stress studies.Plant Soil, 39: 205-207. |
| 9 | Benamar A, Tallon C, Macherel D.2003. Membrane integrity and oxidative properties of mitochondria isolated from imbibing pea seeds after priming or accelerated ageing.Seed Sci Res, 13: 35-45. |
| 10 | Cohen Y R.2002. ß-aminobutyric acid-induced resistance against plant pathogens.Plant Dis, 86(5): 448-457. |
| 11 | Cohen Y, Rubin A E, Kilfin G.2010. Mechanisms of induced resistance in lettuce against Bremia lactucae by DL-β-amino-butyric acid (BABA).Eur J Plant Pathol, 126: 553-573. |
| 12 | Cushman J C.2001. Osmoregulation in plants: Implications for agriculture.Amer Zool, 41: 758-769. |
| 13 | Dat J, Vandenabeele S, Vranova E, van Montagu M, Inzé D, van Breusegem F.2000. Dual action of the active oxygen species during plant stress responses.Cell Mol Life Sci, 57(5): 779-795. |
| 14 | Dubois M, Gilles K A, Hamilton J K, Rebers P A, Smith F.1956. Colorimetric method for determination of sugars and related substances.Anal Chem, 28(3): 350-356. |
| 15 | Ella E S, Dionisio-Sese M L, Ismail A M.2011. Seed pre-treatment in rice reduces damage, enhances carbohydrate mobilization and improves emergence and seedling establishment under flooded conditions. AoB Plants: plr007. |
| 16 | Farhoudi R, Saeedipour S, Mohammadreza D.2011. The effect of NaCl seed priming on salt tolerance, antioxidant enzyme activity, proline and carbohydrate accumulation of muskmelon (Cucumis melo L.) under saline condition.Afr J Agric Res, 6(6): 1363-1370. |
| 17 | Farooq M, Basra S M A, Hafeez K.2006. Seed invigoration by osmohardening in fine and course rice.Seed Sci Technol, 34(1): 181-187. |
| 18 | Fatima R A, Ahmad M.2004. Certain antioxidant enzymes of Allium cepa as biomarkers for the detection of toxic heavy metals in waste water.Sci Total Environ, 346: 256-273. |
| 19 | Gangopadhyay G, Basu S.2000. Proline as osmotic stress marker in in vitro system. In: Hemantaranjan A. Plant Physiology, Biochemistry and Plant Molecular Biology. 3rd edn. Scientific Publishers, Jodhpur, India: 283-304. |
| 20 | Gaspar T, Penel C, Greppin H.1975. Peroxidase and isoperoxidase in relation to root and flower formation.Plant Biochem J, 2: 33-47. |
| 21 | Giannopolitis C N, Reis S K.1977. Superoxide dismutase: I. Occurance in higher plants.Plant Physiol, 59(2): 309-314. |
| 22 | Giorgini J F, Campos C A S P.1992. Changes in the content of soluble sugars and starch synthesis and degradation during germination and seedling growth of Coffea arabica L.Rev Bras Fisiol Vegetal, 4(1): 11-15. |
| 23 | Goswami A, Banerjee R, Raha S.2013. Drought resistance in rice seedlings conferred by seed priming: Role of the anti-oxidant defense mechanisms.Protoplasma, 250: 1115-1129. |
| 24 | Hageman R H, Reed A J.1980. Nitrate reductase from higher plants. In: Sanpietra A. Methods in Enzymology. New York, USA: Academic: 491-503. |
| 25 | Heath R L, Packer L.1968. Photoperoxidation in isolated chloroplasts: I. Kinetics and stochiometry of fatty acid peroxidation.Pak J Bot, 125(1): 189-198. |
| 26 | Hodge S, Thompson G A, Powell G.2005. Application of DL-beta-aminobutyric acid (BABA) as a root drench to legumes inhibits the growth and reproduction of the pea aphid Acyrthosiphon pisum (Hemiptera: Aphididae).Bull Entomol Res, 95(5): 449-455. |
| 27 | Jakab G, Ton J, Flors V, Zimmerli L, Metraux J P, Mauch-Mani B.2005. Enhancing Arabidopsis salt and drought stress tolerance by chemical priming for its abscisic acid responses.Plant Physiol, 139(1): 267-274. |
| 28 | Jisha K C, Vijayakumari K, Puthur J T.2013. Seed priming for abiotic stress tolerance: An overview.Acta Physiol Plant, 35(5): 1381-1396. |
| 29 | Jisha K C, Puthur J T.2014. Halopriming of seeds imparts tolerance to NaCl and PEG induced stress in Vigna radiata (L.) Wilczek varieties.Physiol Mol Biol Plants, 20(3): 303-312. |
| 30 | Kameli A, Losel D M.1993. Carbohydrates and water status in wheat plants under water stress.New Phytol, 125(3): 609-614. |
| 31 | Kavi Kishore P B, Sangam S, Amrutha R N, Sri Laxmi P, Naidu K R, Rao K R S S, Rao S, Reddy K J, Theriappan P, Sreenivasulu N.2005. Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: Its implications in plant growth and abiotic stress tolerance.Curr Sci, 88: 424-438. |
| 32 | Keyvan S.2010. The effects of drought stress on yield, relative water content, proline, soluble carbohydrates and chlorophyll of bread wheat cultivars.J Anim Plant Sci, 8(3): 1051-1060. |
| 33 | Kolloffel C.1967. Respiration rate and mitochondrial activity in the cotyledons of Pisum sativum during germination.Acta Bot Neerl, 16(3): 111-122. |
| 34 | Lara T S, Lira J M S, Rodrigues A C, Rakocevic M, Alvarenga A A.2014. Potassium nitrate priming affects the activity of nitrate reductase and antioxidant enzymes in tomato germination.J Agric Sci, 6(2): 72-80. |
| 35 | Lowry O H, Rosenbrough N J, Farr A L, Randall R J.1951. Protein measurement with folin-phenol reagent.J Biol Chem, 193: 265-275. |
| 36 | Macovei A, Garg B, Raikwar S, Balestrazzi A, Carbonera D, Buttafava A, Bremont J F J, Gill S S, Tuteja N. 2014. Synergistic exposure of rice seeds to different doses of γ-ray and salinity stress resulted in increased antioxidant enzyme activities and gene-specific modulation of TC-NER pathway.Biol Med Res Int, 2014: 1-15. |
| 37 | Mathew J, Mohanasarida K.2005. Seed priming on crop establishment and seedling vigour in semi-dry rice (Oryza sativa).Res Crops, 6: 23-25. |
| 38 | Mayer A, Eskandari S, Grallath S, Rentsch D.2006. AtGAT1, a high affinity transporter for γ-aminobutyric acid in Arabidopsis thaliana. J Biol Chem, 281: 7197-7204. |
| 39 | Mittler R.2002. Oxidative stress, antioxidants and stress tolerance.Trends Plant Sci, 7: 405-410. |
| 40 | Mondal S, Viji P, Bose B.2011. Role of seed hardening in rice variety Swarna (MTU 7029).Res J Seed Sci, 4(3): 157-165. |
| 41 | Moosavi A, Afshari R T, Sharif-Zadeh F, Aynehband A.2009. Effect of seed priming on germination characteristics, polyphenoloxidase, and peroxidase activities of four amaranth cultivars. J Food Agric Environ, 7: 353-358. |
| 42 | Nawaz A, Amjad M, Pervez M A, Afzal I.2011. Effect of halopriming on germination and seedling vigor of tomato.Afr J Agric Res, 6: 3551-3559. |
| 43 | Nawaz F, Ashraf M Y, Ahmad R, Waraich E A.2013. Selenium (Se) seed priming induced growth and biochemical changes in wheat under water deficit conditions.Biol Trace Element Res, 151(2): 284-293. |
| 44 | Nouman W, Siddiqui M T, Basra S M A, Afzal I, Rehman H U.2012. Enhancement of emergence potential and stand establishment of Moringa oleifera Lam. by seed priming.Turk J Agric Forest, 36: 227-235. |
| 45 | Oka Y, Cohen Y, Spiegel Y.1999. Local and systemic induced resistance to the root-knot nematode in tomato by DL-β-amino-n-butyric acid.Phytopathology, 89: 1138-1143. |
| 46 | Oukarroum A, Schansker G, Strasser R J.2009. Drought stress effects on photosystem I content and photosystem II thermo tolerance analyzed using Chl a fluorescence kinetics in barley varieties differing in their drought tolerance.Physiol Plant, 137(2): 188-199. |
| 47 | Patade V Y, Bhargava S, Suprasanna P.2009. Halopriming imparts tolerance to salt and PEG induced drought stress in sugarcane.Agric Ecosyst Environ, 134: 24-28. |
| 48 | Puthur J T.2000. Photosynthetic events in Sesbania sesban (L.) Merrill in relation to osmotic stress during different developmental stages. Ph.D. Thesis, New Delhi, India. |
| 49 | Ruan S, Xue Q, Tylkowska R.2002. Effects of priming on seed germination and health of rice (Oryza sativa L.) seeds.Seed Sci Technol, 30: 451-458. |
| 50 | Sass R L, Cicerone R J.2002. Photosynthate allocations in rice plants: Food production or atmospheric methane?J Plant Physiol, 99: 11993-11995. |
| 51 | Scandalios J G.1993. Oxygen stress and superoxide dismutases.Plant Physiol, 101(1): 7-12. |
| 52 | Schmitt N, Dizengremel P.1989. Effect of osmotic stress on mitochondria isolated from etiolated mung bean and sorghum seedlings.Plant Physiol Biochem, 27: 17-26. |
| 53 | Sharma S S, Dietz K J.2006. The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress.J Exp Bot, 57: 711-726. |
| 54 | Sharma A D, Rathore S V S, Srinivasana K, Tyagia R K.2014. Comparison of various seed priming methods for seed germination, seedling vigour and fruit yield in okra (Abelmoschus esculentus L. Moench).Scient Hort, 165: 75-81. |
| 55 | Singh-Tomar R, Mathur S, Allakhverdier S I, Jajoo A.2012. Changes in PS II heterogeneity in response to osmotic and ionic stress in wheat leaves (Triticum aestivum).J Bioenerg Biomembr, 44: 411-419. |
| 56 | Strasser R J, Srivastava A, Tsmilli-Michael M.2004. Analysis of the chlorophyll a fluorescence transient. In: Papageorgiou G C, Govindjee. Chlorophyll a Fluorescence: A Signature of Photosynthesis. Dordrechet: Springer: 321-362. |
| 57 | Sudharani M, Reddy P R, Jayalakshmi V.2012. A comprehensive review on ‘genetic components of salinity tolerance in rice (Oryza sativa L.).’Int J Appl Biol Pharm Technol, 3: 312-322. |
| 58 | Sun Y Y, Sun Y J, Wang M T, Li X Y, Guo X, Hu R, Ma J.2010. Effects of seed priming on germination and seedling growth under water stress in rice.Acta Agron Sin, 36: 1931-1940. (in Chinese with English abstract) |
| 59 | Tabrizi E F M, Yarnia M, Ahmadzadeh V, Farjzadeh N.2011. Priming effect of different times of maize seeds with nutrient elements in water stress on corn yield.Ann Biol Res, 2(3): 419-423. |
| 60 | Ton J, Jakab G, Toquin V, Flors V, Iavicoli A, Maeder M N, Metraux J P, Mauch-Mani B.2005. Dissecting the β-amino butyric acid-induced priming phenomenon in Arabidopsis.Plant Cell, 17(3): 987-999. |
| 61 | Varier A, Vari A K, Dadlani M.2010. The subcellular basis of seed priming.Curr Sci, 99: 450-456. |
| 62 | Wang W, Vinocur B, Altman A.2003. Plant responses to drought, salinity and extreme temperatures: Towards genetic engineering for stress tolerance.Planta, 218(1): 1-14. |
| 63 | Wang W, Vinocur B, Shoseyov O, Altman A.2004. Role of plant heat shock proteins and molecular chaperones in the abiotic stress response.Trends Plant Sci, 9(5): 244-252. |
| 64 | Worrall D, Holrod G H, Moore J P, Glowacz M, Croft P, Taylor J E, Paul N D, Roberts M R.2012. Treating seeds with activators of plant defence generates long-lasting priming of resistance to pests and pathogens.New Phytol, 193: 770-778. |
| 65 | Yang C M, Sung C M.1980. Relations between nitrate reductase activity and growth of rice seedlings.J Agric Assoc China, 111: 15-23. |
| 66 | Zhong Y P, Wang B, Yan J H, Cheng L J, Yao L M, Xiao L, Wu T L.2014. DL-β-amino butyric acid induced resistance in soybean against Aphis glycines Matsumura (Hemiptera: Aphididae).PLoS One, 9(1): 1-11. |
| 67 | Zimmerli L, Hou B H, Tsai C H, Jakab G, Mauch-Mani B, Somerville S.2008. The xenobiotic beta-amino butyric acid enhances Arabidopsis thermo-tolerance.Plant J, 53(1): 144-156. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [13] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||