
Rice Science ›› 2016, Vol. 23 ›› Issue (1): 42-50.DOI: 10.1016/j.rsci.2016.01.005
• Orginal Article • Previous Articles Next Articles
Rahman Moni Zakiah1, Ansar Ali Md2, Shahidul Alam Md3, Asif Rahman Md4, Rejwan Bhuiyan Md4,5, Salim Mian Md4,5, Md Iftekharuddaula Khandakar1, Abdul Latif Md4,5, Ashik Iqbal Khan Mohammad4,5( )
)
Received:2015-06-18
Accepted:2015-08-18
Online:2016-01-20
Published:2015-11-05
Rahman Moni Zakiah, Ansar Ali Md, Shahidul Alam Md, Asif Rahman Md, Rejwan Bhuiyan Md, Salim Mian Md, Md Iftekharuddaula Khandakar, Abdul Latif Md, Ashik Iqbal Khan Mohammad. Morphological and Genetical Variability among Rhizoctonia solani Isolates Causing Sheath Blight Disease of Rice[J]. Rice Science, 2016, 23(1): 42-50.
Add to citation manager EndNote|Ris|BibTeX
| Code | Location | Agro-ecological zone |
| GA1, GA2, GA3, GA4, GA5 | Gazipur | Madhupur tract |
| GO1, GO2, GO3, GO4, GO5 | Godagari | High ganger river flood plain |
| TA1, TA2, TA3, TA4, TA5 | Tanor | High ganger river flood plain |
| CO1, CO2, CO3 | Comilla | Middle Meghna river flood plan |
Table 1 List of R. solani isolates with geographic locations.
| Code | Location | Agro-ecological zone |
| GA1, GA2, GA3, GA4, GA5 | Gazipur | Madhupur tract |
| GO1, GO2, GO3, GO4, GO5 | Godagari | High ganger river flood plain |
| TA1, TA2, TA3, TA4, TA5 | Tanor | High ganger river flood plain |
| CO1, CO2, CO3 | Comilla | Middle Meghna river flood plan |
| Character | Character state | ||
| Mycelial color on potato dextrose agar, color of sclerotia | 0, hyaline; 1, cream or faint brown; 2, light brown; 3, medium brown; 4, dark brown | ||
| Superficial sclerotia (SS) dispersed on whole colony, exudate droplets on sclerotium surface | 0, present; 1, absent | ||
| Colony reverse: pigment | 0, not present; 1, cream or faint brown; 2, light brown | ||
| Sclerotia on lid | 0, absent; 1, present | ||
| Topography of sclerotia | 0, immersed; 1, superficial | ||
| Shape of sclerotia | 0, flattened bottom and round top; 1, superficial; 2, irregularly globose with pitted surface; 3, irregular | ||
| Arial mycelial quality | 0, absent; 1, all hyphae close to surface of agar; 2, air space in dish half filled; 3, almost all airspace filled | ||
| Quantity of sclerotia, pseudo sclerotia, SS discrete, SS aggregated, SS scattered, SS near inoculum, SS near margin, dark brown runner hyphae in aerial mycelium and/or on colony surface, growth on lid | 0, absent; 1, few; 2, moderate; 3, abundant | ||
Table 2. Morphological characteristics description of R. solani isolates and their attributes.
| Character | Character state | ||
| Mycelial color on potato dextrose agar, color of sclerotia | 0, hyaline; 1, cream or faint brown; 2, light brown; 3, medium brown; 4, dark brown | ||
| Superficial sclerotia (SS) dispersed on whole colony, exudate droplets on sclerotium surface | 0, present; 1, absent | ||
| Colony reverse: pigment | 0, not present; 1, cream or faint brown; 2, light brown | ||
| Sclerotia on lid | 0, absent; 1, present | ||
| Topography of sclerotia | 0, immersed; 1, superficial | ||
| Shape of sclerotia | 0, flattened bottom and round top; 1, superficial; 2, irregularly globose with pitted surface; 3, irregular | ||
| Arial mycelial quality | 0, absent; 1, all hyphae close to surface of agar; 2, air space in dish half filled; 3, almost all airspace filled | ||
| Quantity of sclerotia, pseudo sclerotia, SS discrete, SS aggregated, SS scattered, SS near inoculum, SS near margin, dark brown runner hyphae in aerial mycelium and/or on colony surface, growth on lid | 0, absent; 1, few; 2, moderate; 3, abundant | ||
| Primer | Sequence | ||
| Variable number of tandem repeat (VNTR) | |||
| MR | GAGGGTGGCGGTTCT | ||
| RY | CAGCAGCAGCAGCAG | ||
| GF | TCCTCCTCCTCCTCC | ||
| Amplified fragment length polymorphisms (AFLP) | |||
| AFLP-C | GACTAGGATACATGCAGGC | ||
| AFLP-D | GACTACGTACATGKACKGKAC | ||
Table 3. List of primers and their sequences.
| Primer | Sequence | ||
| Variable number of tandem repeat (VNTR) | |||
| MR | GAGGGTGGCGGTTCT | ||
| RY | CAGCAGCAGCAGCAG | ||
| GF | TCCTCCTCCTCCTCC | ||
| Amplified fragment length polymorphisms (AFLP) | |||
| AFLP-C | GACTAGGATACATGCAGGC | ||
| AFLP-D | GACTACGTACATGKACKGKAC | ||
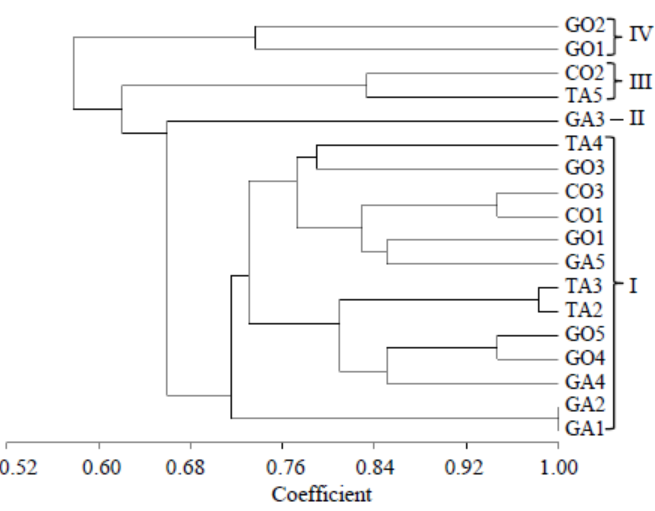
Fig. 2. Un-weighted pair group method with arithmetic mean dendrogram of R. solani isolates constructed with Multi Variaty Statistical Package ver. 3.1 using the Gower’s general similarity coefficient based on morphological characters.
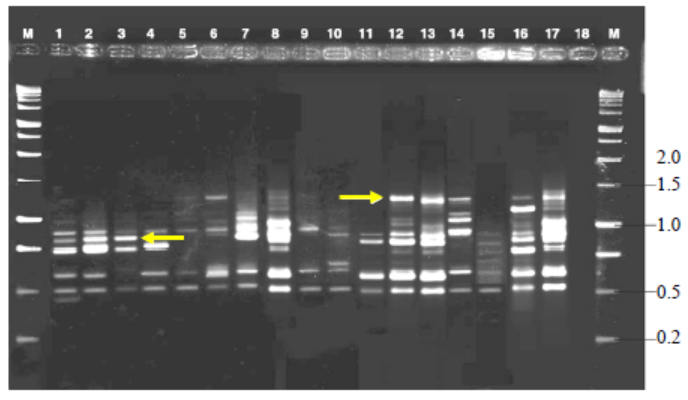
Fig. 3. Variable number of tandem repeat fingerprinting patterns of total genomic DNA of R. solani isolates with primer MR.M, Kilo base (kb) ladder; Lanes 1 to 18, Isolates GA1, GA2,GA3, GA4, GA5, GO1, GO2, GO3, GO4, GO5, TA1, TA2, TA3, TA4,TA5, CO1, CO2 and CO3, respectively.Arrow indicates polymorphic bands.
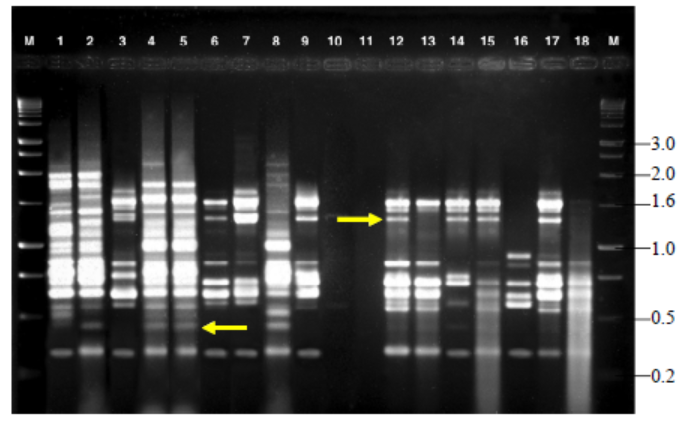
Fig. 4. Variable number of tandem repeat fingerprinting patterns of total genomic DNA of R. solani isolates with primer RY. M, Kilo base (kb) ladder; Lanes 1 to 18, Isolates GA1, GA2, GA3, GA4, GA5, GO1, GO2, GO3, GO4, GO5, TA1, TA2, TA3, TA4, TA5, CO1, CO2 and CO3, respectively.Arrow indicates polymorphic bands.
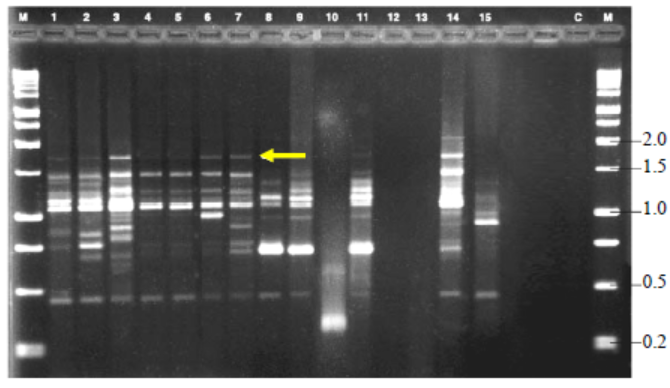
Fig. 5. Variable number of tandem repeat fingerprinting patterns of total genomic DNA of R. solani isolates with primer GF.M, Kilo base (kb) ladder; Lanes 1 to 18, Isolates GA1, GA2, GA3,GA4, GA5, GO1, GO2, GO3, GO4, GO5, TA1, TA2, TA3, TA4, TA5,CO1, CO2 and CO3, respectively.Arrow indicates polymorphic bands.
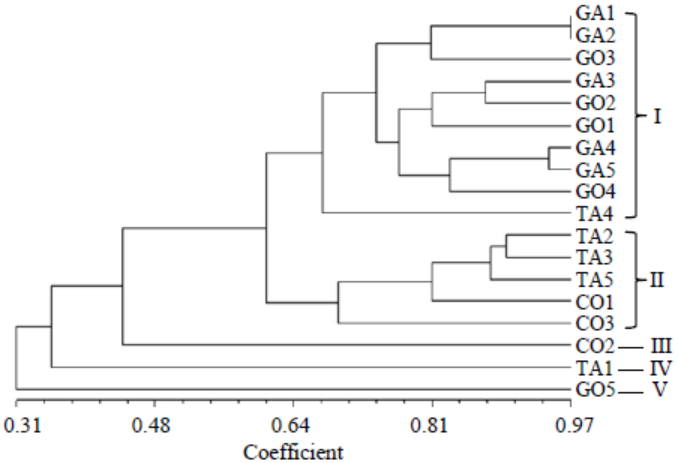
Fig. 6. Un-weighted pair group method with arithmetic mean dendrogram of R. solani isolates constructed with Multi Variety Statistical Package ver 3.1 using the Gower’s general similarity coefficient based on DNA fingerprinting (variable number of tandem repeat analysis of primers MR, RY, and GF).
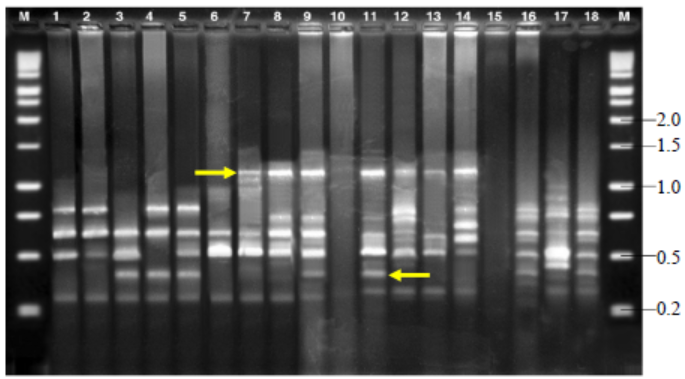
Fig. 7. Amplified fragment length polymorphisms fingerprinting patterns of total genomic DNA of R. solani isolates with primer AFLP-C.M, Kilo base (kb) ladder; Lanes 1 to 18, Isolates GA1, GA2, GA3, GA4, GA5, GO1, GO2, GO3, GO4, GO5, TA1, TA2, TA3, TA4, TA5, CO1, CO2 and CO3, respectively.Arrow indicates polymorphic bands.
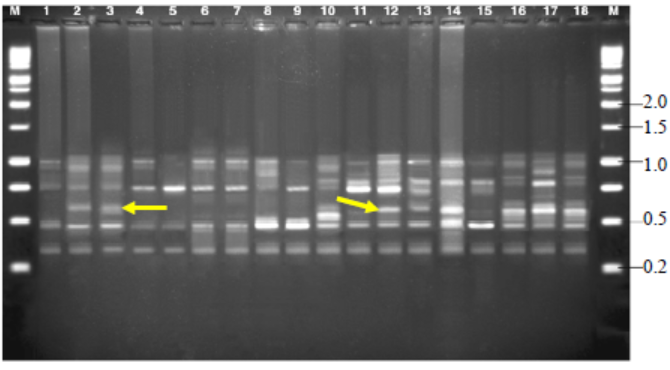
Fig. 8. Amplified fragment length polymorphisms fingerprinting patterns of total genomic DNA of R. solani isolates with primer AFLP-D.M, Kilo base (kb) ladder; Lanes 1 to 18, Isolates GA1, GA2, GA3,GA4, GA5, GO1, GO2, GO3, GO4, GO5, TA1, TA2, TA3, TA4, TA5,CO1, CO2 and CO3, respectively.
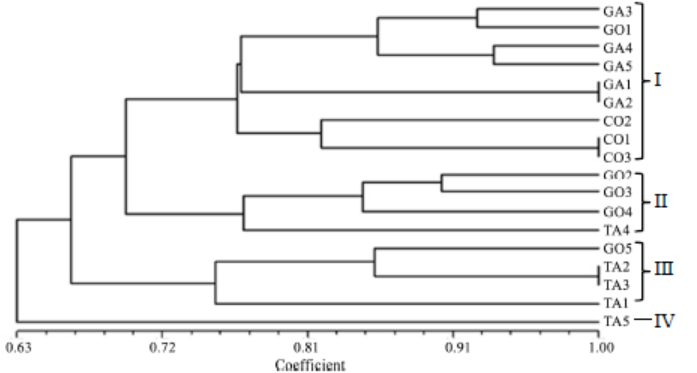
Fig. 9. Un-weighted pair group method with arithmetic mean dendrogram of relatedness of R. solani AG1 IA isolates constructed with Multi Variaty Statistical Package ver 3.1 using the Gower’s general similarity coefficient based on amplified fragment length polymorphism analysis of primers AFLP-C and AFLP-D.
| [1] | Ali M A.2002. Biological Variation and Chemical Control of Rhizoctnia solani Causing Rice Sheath Blight in Bangladesh. Department of Biological Sciences, Imperial College for Science, Technology and Medicine. Silwod Park, Ascot, Berkshire: 202. |
| [2] | Banerjee S, Datta S, Mondal A, Bhattacharya S.2012. Characterization of molecular variability in Rhizoctonia solani isolate from different agro-ecological zone by random amplified polymorphic DNA (RAPD) markers.Afr J Biotechnol, 11(40): 9543-9548. |
| [3] | Banniza S, Rutherford M A.2001. Diversity of isolates of Rhizoctonia solani AG-1 IA and their relationship to other anastomosis groups based on pectic zymograms and molecular analysis.Mycol Res, 105(1): 33-40. |
| [4] | Basu A, Gupta P K S.1992. Loss in yield and seed infection in promising genotypes of rice (Oryza sativa) due to sheath blight disease caused by Rhizoctonia solani.Ind J Agric Sci, 62(8): 570-571. |
| [5] | Bonman J M.1992. Durable resistance to rice blast disease: Environment influences.Euphytica, 63: 115-123. |
| [6] | Bruns T D, White T J, Taylor J W.1991. Fungal molecular systematics.Ann Rev Ecol Syst, 22: 525-564. |
| [7] | Flor H H.1971. Current status of the gene-for-gene concept.Ann Rev Phytopathol, 9: 275-296. |
| [8] | Jeffreys A J, Wilson V, Thien S L.1985a. Hyper-variable mini-satellite regions in human DNA.Nature, 314: 67-73. |
| [9] | Jeffreys A J, Wilson V, Thien S L.1985b. Individual specific fingerprints of human DNA.Nature, 316: 76-79. |
| [10] | Julian M C, Acero J, Salazar O, Keijer J, Rubio V.1999. Mating type correlated molecular markers and demonstration of heterokaryosis in the phytopathogenic fungus Thanatephorus cucumeris (Rhizoctonia solani) AG1 1C by AFLP DNA fingerprinting analysis.J Biotechnol, 67(1): 49-56. |
| [11] | Khodayari M, Safaie N, Shamsbakhsh M.2009. Genetic diversity of Iranian AG1-IA isolates of Rhizoctonia solani, the cause of rice sheath blight, using morphological and molecular markers.J Phytopathol, 157: 708-714. |
| [12] | Majer D, Mithen R, Lewis B G, Vos P, Oliver R P.1996. The use of AFLP fingerprinting for detection of genetic variation fungi.Mycol Res, 100(9): 1107-1111. |
| [13] | Matsumoto M, Furuya N, Matsuyama N.1996. PCR-RFLP analysis of amplified 28S ribosomal DNA for identification of Rhizoctonia spp., the causal agents of sheath diseases of rice plants.J Fac, 41: 39-44. |
| [14] | Mekwatanakarn P, Kositratana W, Phromraksa T, Zeigler R S.1999. Sexually fertile Magnaporthe grisea rice pathogens in Thailand.Plant Dis, 83(10): 939-943. |
| [15] | Mian M S, Stevens C, Mia M A T.2003. Diversity of the rice blast pathogen Pyricularia grisea from Bangladesh analysed by DNA fingerprinting.Bangl J Plant Pathol, 1: 81-85. |
| [16] | Nakamura Y, Leppert M, Connel P, Wolf R, Holm T, Culver M, Martin C, Fujimoti E, Holf M, Kumalin E, White R.1987. Variable number of tandem repeat (VNTR) marker for human gene mapping.Science, 235: 1616-1622. |
| [17] | Ogoshi A.1987. Ecology and pathogenicity of anastomosis and intra-specific groups of Rhizoctonia solani Kuhn.Ann Rev Phytopathol, 25: 125-143. |
| [18] | Parmeter J R, Sherwood R T, Platt W D.1969. Anastomosis grouping among isolates of Thanatephorus cucumaris. Phytopathology, 59: 1270-1278. |
| [19] | Parmeter J R, Whitney H S.1970. Taxonomy and nomenclature of the imperfect state. In: Parmeter J R. Rhizoctonia solani: Biology and Pathology. Berkely: University of California Press: 7-19. |
| [20] | Pascual C B, Toda T, Raymondo A D, Hyakumachi M.2000. Characterization by convential techiques and PCR of Rhizoctonia solani isolates causing banded leaf sheath blight in maize.Plant Pathol, 49: 108-118. |
| [21] | Raeder U, Broda P.1985. Rapid preparation of DNA from filamentous fungi.Lett Appl Microbiol, 1: 17-20. |
| [22] | Rashad Y M, Abdel-Fattah G M, Hafez E E, El-Haddad S A.2012. Diversity among some Egyptian isolates of Rhizoctonia solani based on anastomosis grouping, molecular identification and virulence on common bean.Afr J Microbiol Res, 6(37): 6661-6667. |
| [23] | Rohlf F J.2000. NTSYS-pc. Numerical Taxonomy and Multivariate System version 2.11a. Exeter Software, Setauket, New York, USA. |
| [24] | Sambrook J, Russel D W.2001.Molecular Cloning: A Laboratory Manual. 3rd edn. New York: Cold Spring Harbor Laborary Press. |
| [25] | Sharma M, Gupta S K, Sharma T R.2005. Characterization of variability in Rhizoctonia solani by using morphological and molecular markers.Phytopathology, 153: 449-456. |
| [26] | Sharma N R, Akanda S I, Shahjahan A K M.1995. Development of sheath blight in short, tall, early and late maturing rice cultivars.Bangl J Bot, 24(2): 143-146. |
| [27] | Sherwood R T.1969. Morphology and pathology in four anastomosis groups ofThanatephorus cucumeris. J Phytopathol, 59: 1924-1929. |
| [28] | Silue D, Tharreau D, Notteghem J L.1992. Evidence of gene-for-gene relationship in the Oryza sativa-Magnaporthe grisea pathosystem.Phytopathology, 82: 577-580. |
| [29] | Singh A, Singh U S, Willocquet L, Savary S.1999. Relationship among cultural morphological characteristics, anastomosis behavior and pathogenicity of Rhizoctonia solani Khun on rice.J Mycol Plant Pathol, 29: 306-316. |
| [30] | Singh V, Singh U S, Singh K P, Singh M, Kumar A.2002. Genetic diversity of Rhizoctonia solani isolated from rice: Differentiation by morphological characteristics, pathogenecity, anastomosis behaviour and RAPD fingerprinting.J Mycol Plant Pathol, 32(3): 332-344. |
| [31] | Sivalingan P N, Vishwakarma S N, Singh U S.2006. Role of seed-borne inoculum of Rhizoctonia solani in sheath blight of rice.Ind J Phytopath, 59(4): 445-452. |
| [32] | Sneh B, Burpee L, Ogoshi A.1991. Identification of Rhizoctonia species. St Paul, Minnesota: APS Press. |
| [33] | Tajick M A, Rahimianm H, Alizadeh A.2005. Studies on population of Rhizoctonia solani AG 1 1A isolated from rice by rDNA RFLP in Mazandaran Province.Iran J Plant Pathol, 41: 507-542. |
| [34] | Thakur R S, Sugha S K, Sharma B M.1992. Morphological grouping of different isolates of Rhizoctonia solani Khun.Plant Dis Res, 7: 58-59. |
| [35] | Toda T, Nasu H K, Kageyama K, Hyakumachi M.1998. Genetic identification of web-blight (Rhizoctonia solani AGI) obtained from European pear using RFLP of rDNA-ITS and RAPD analysis.Res Bull Fac Agric Gifu Univ, 63: 1-9. |
| [36] | Vijayam M, Chandrasekharan N M.1985. Anastomosis grouping of isolates of Rhizoctonia solani Kuha (Thanatephorus cucumeris (Frank) Donk) causing sheath blight of rice.Curr Sci, 54(6): 289-291. |
| [37] | Zhou E, Jia Y, Singh P, Correll J C, Lee F N.2007. Instability of the Magnaporthe oryzaea virulence gene AVR-Pitai alters virulence.Fungal Genet Biol, 44: 1024-1034. |
| [1] | Prathap V, Suresh KUMAR, Nand Lal MEENA, Chirag MAHESHWARI, Monika DALAL, Aruna TYAGI. Phosphorus Starvation Tolerance in Rice Through a Combined Physiological, Biochemical and Proteome Analysis [J]. Rice Science, 2023, 30(6): 8-. |
| [2] | Serena REGGI, Elisabetta ONELLI, Alessandra MOSCATELLI, Nadia STROPPA, Matteo Dell’ANNO, Kiril PERFANOV, Luciana ROSSI. Seed-Specific Expression of Apolipoprotein A-IMilano Dimer in Rice Engineered Lines [J]. Rice Science, 2023, 30(6): 6-. |
| [3] | Sundus ZAFAR, XU Jianlong. Recent Advances to Enhance Nutritional Quality of Rice [J]. Rice Science, 2023, 30(6): 4-. |
| [4] | Kankunlanach KHAMPUANG, Nanthana CHAIWONG, Atilla YAZICI, Baris DEMIRER, Ismail CAKMAK, Chanakan PROM-U-THAI. Effect of Sulfur Fertilization on Productivity and Grain Zinc Yield of Rice Grown under Low and Adequate Soil Zinc Applications [J]. Rice Science, 2023, 30(6): 9-. |
| [5] | FAN Fengfeng, CAI Meng, LUO Xiong, LIU Manman, YUAN Huanran, CHENG Mingxing, Ayaz AHMAD, LI Nengwu, LI Shaoqing. Novel QTLs from Wild Rice Oryza longistaminata Confer Rice Strong Tolerance to High Temperature at Seedling Stage [J]. Rice Science, 2023, 30(6): 14-. |
| [6] | LIN Shaodan, YAO Yue, LI Jiayi, LI Xiaobin, MA Jie, WENG Haiyong, CHENG Zuxin, YE Dapeng. Application of UAV-Based Imaging and Deep Learning in Assessment of Rice Blast Resistance [J]. Rice Science, 2023, 30(6): 10-. |
| [7] | Md. Forshed DEWAN, Md. AHIDUZZAMAN, Md. Nahidul ISLAM, Habibul Bari SHOZIB. Potential Benefits of Bioactive Compounds of Traditional Rice Grown in South and South-East Asia: A Review [J]. Rice Science, 2023, 30(6): 5-. |
| [8] | Raja CHAKRABORTY, Pratap KALITA, Saikat SEN. Phenolic Profile, Antioxidant, Antihyperlipidemic and Cardiac Risk Preventive Effect of Chakhao Poireiton (A Pigmented Black Rice) in High-Fat High-Sugar induced Rats [J]. Rice Science, 2023, 30(6): 11-. |
| [9] | LI Qianlong, FENG Qi, WANG Heqin, KANG Yunhai, ZHANG Conghe, DU Ming, ZHANG Yunhu, WANG Hui, CHEN Jinjie, HAN Bin, FANG Yu, WANG Ahong. Genome-Wide Dissection of Quan 9311A Breeding Process and Application Advantages [J]. Rice Science, 2023, 30(6): 7-. |
| [10] | JI Dongling, XIAO Wenhui, SUN Zhiwei, LIU Lijun, GU Junfei, ZHANG Hao, Tom Matthew HARRISON, LIU Ke, WANG Zhiqin, WANG Weilu, YANG Jianchang. Translocation and Distribution of Carbon-Nitrogen in Relation to Rice Yield and Grain Quality as Affected by High Temperature at Early Panicle Initiation Stage [J]. Rice Science, 2023, 30(6): 12-. |
| [11] | Nazaratul Ashifa Abdullah Salim, Norlida Mat Daud, Julieta Griboff, Abdul Rahim Harun. Elemental Assessments in Paddy Soil for Geographical Traceability of Rice from Peninsular Malaysia [J]. Rice Science, 2023, 30(5): 486-498. |
| [12] | Tan Jingyi, Zhang Xiaobo, Shang Huihui, Li Panpan, Wang Zhonghao, Liao Xinwei, Xu Xia, Yang Shihua, Gong Junyi, Wu Jianli. ORYZA SATIVA SPOTTED-LEAF 41 (OsSPL41) Negatively Regulates Plant Immunity in Rice [J]. Rice Science, 2023, 30(5): 426-436. |
| [13] | Monica Ruffini Castiglione, Stefania Bottega, Carlo Sorce, Carmelina SpanÒ. Effects of Zinc Oxide Particles with Different Sizes on Root Development in Oryza sativa [J]. Rice Science, 2023, 30(5): 449-458. |
| [14] | Ammara Latif, Sun Ying, Pu Cuixia, Noman Ali. Rice Curled Its Leaves Either Adaxially or Abaxially to Combat Drought Stress [J]. Rice Science, 2023, 30(5): 405-416. |
| [15] | Liu Qiao, Qiu Linlin, Hua Yangguang, Li Jing, Pang Bo, Zhai Yufeng, Wang Dekai. LHD3 Encoding a J-Domain Protein Controls Heading Date in Rice [J]. Rice Science, 2023, 30(5): 437-448. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||